Advertisements
Advertisements
प्रश्न
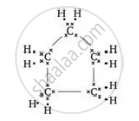
What will be the formula and electron dot structure of cyclopentane?
उत्तर
The formula for cyclopentane is \[\ce{C5H10}\]. Its electron dot structure is given below.

APPEARS IN
संबंधित प्रश्न
‘A’ is an element having four electrons in its outermost orbit. An allotropes ‘B’ of this element is used as a dry lubricant in machinery and in pencil leads. So:
i. Write the name of element ‘A’ and its allotropes.
ii. State whether ‘B’ is a good conductor or non-conductor of electricity?
Define Unsaturated hydrocarbon.
Explain in brief two main reasons for carbon forming a large number of compounds.
Draw the electron dot structures for propanone.
Why are certain compounds called hydrocarbons? Write the general formula for homologous series of alkanes, alkenes and alkynes and also draw the structure of the first member of each series. Write the name of the reaction that converts alkenes into alkanes and also write a chemical equation to show the necessary conditions for the reaction to occur.
Write a short note on Catenation.
Fill in the following blank with suitable word:
Ethyne has ......... carbon-hydrogen single bonds.
Give the names and structural formulae of one saturated cyclic hydrocarbon and one unsaturated cyclic hydrocarbon.
Out of the following pairs of compounds, the unsaturated compounds are:
(a) C2H6 and C4H6
(b) C6H12 and C5H12
(c) C4H6 and C6H12
(d) C2H6 and C4H10
An unsaturated hydrocarbon having a triple covalent bond has 50 hydrogen atoms in its molecule. The number of carbon atoms in its molecule will be:
(a) 24
(b) 25
(c) 26
(d) 28
Write the molecular formula of an alkene having 20 carbon atoms?
Which of the following hydrocarbons is unsaturated?
C3H4; C2H6
What happens when vegetable oils are hydrogenated? Name the catalyst used.
A mixture of ethyne (acetylene) and oxygen is burnt for welding. Can you tell why a mixture of ethyne and air is not used?
Structural formula of ethyne is ______.
Pentane has the molecular formula C5 H12 It has
Which among the following are unsaturated hydrocarbons?
- \[\ce{H3C - CH2 - CH2 - CH3}\]
- \[\ce{H3C - C ≡ C - CH3}\]
- \[\begin{array}{cc}
\ce{H3C - CH - CH3}\\
|\phantom{..}\\
\ce{CH3}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H3C - C = CH2}\\
|\\
\phantom{...}\ce{CH3}\\
\end{array}\]
Identify and name the functional groups present in the following compounds.
- \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{..}\\
\ce{H - C - C - C - OH}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{..}\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}\\
\end{array}\] - \[\begin{array}{cc}
\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{....}\\
\phantom{...}|\phantom{....}|\phantom{....}||\phantom{....}\\
\ce{H - C - C - C - OH}\\
\phantom{...}|\phantom{....}|\phantom{.........}\\
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{.......}\\
\end{array}\] - \[\begin{array}{cc}
\phantom{......}\ce{H}\phantom{....}\ce{H}\phantom{....}\ce{O}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.......}\\
\phantom{....}|\phantom{....}|\phantom{....}||\phantom{....}|\phantom{....}|\phantom{....}\\
\ce{H} - \ce{C} - \ce{C} - \ce{C} - \ce{C} - \ce{C} - \ce{H}\\
\phantom{....}|\phantom{.....}|\phantom{........}|\phantom{....}|\phantom{....}\\
\phantom{....}\ce{H}\phantom{....}\ce{H}\phantom{........}\ce{H}\phantom{..}\ce{H}\phantom{.....}\\
\end{array}\] - \[\begin{array}{cc}
\phantom{......}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{......}\\
\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}\\
\ce{H - C - C - C = C - H}\\
\phantom{....}|\phantom{....}|\phantom{.............}\\
\phantom{....}\ce{H}\phantom{...}\ce{H}\phantom{.............}
\end{array}\]
