Advertisements
Advertisements
प्रश्न
Draw the structures for Ethanoic acid.
उत्तर
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{...}\ce{O}\phantom{.}\\
\phantom{.}|\phantom{....}||\phantom{..}\\
\ce{H - C - C - OH}\\
|\phantom{......}\\
\ce{H}\phantom{......}\\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
List two reasons for carbon forming a large number of compounds. Name the type of bonding found in most of its compounds. Why does carbon form compounds mainly by this kind of bonding?
Give reason why the carbon compounds do not conduct electricity in the molten state.
Write the names and structural formulae of all the possible isomers of hexane.
Which of the two has a branched chain : isobutane or normal butane?
Write the molecular formula and structure of cyclohexane. How many covalent bonds are there in a molecule of cyclohexane?
How many isomers of the following hydrocharbons are possible?
C4H10
A diamond-toothed saw is usually used for cutting:
(a) steel girders
(b) logs of wood
(c) marble slabs
(d) asbestos sheets
The number of carbon atoms in the organic compound named as 2,2-dimethylpropane is:
(a) two
(b) five
(c) three
(d) four
Two organic compounds A and B have the same molecular formula C6H12. Write the names and structural formulae:
Which compound contains only single bonds?
You are given the following molecular formulae of some hydrocarbons:
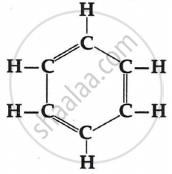
Which formula represents benzene?
Name the functional group which always occurs in the middle of a carbon chain.
Intake of small quantity of methanol can be lethal. Comment.
Catenation is the ability of an atom to form bonds with other atoms of the same element. It is exhibited by both carbon and silicon. Compare the ability of catenation of the two elements. Give reasons.
Recognise the carbon chain type of the following:
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\\ce{H - C - C - C - C - H}\\|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}
\end{array}\]
Write the structures of two compounds having molecular formula C3H6O and give their names.
Draw two structural isomers of butane.
Draw the structure of propanol.
Name the following: