Advertisements
Advertisements
प्रश्न
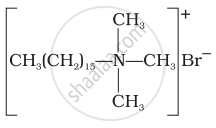
Explain the following terms with suitable examples - Non-ionic detergents
उत्तर १
Non-ionic detergents - Molecules of these detergents do not contain any ions. These detergents are esters of alcohols having high molecular mass. They are obtained by reacting polyethylene glycol and stearic acid.

उत्तर २
Neutral or non-ionic detergents: These are esters of high molecular mass alcohols with fatty acids. These can also be obtained by treatment of long chain alcohols by with excess of ethylene oxide in presence of a base. For example, polyethylene glycol stearate,CH3(CH2)16COO (CH2CH2O)11 CH2CH2OH Polyethylene glycol stearate.
APPEARS IN
संबंधित प्रश्न
Explain the following term with a suitable example:
Cationic detergents
Label the hydrophilic and hydrophobic parts in the following compounds.
CH3(CH2)10CH2OSO3 –Na+
Label the hydrophilic and hydrophobic parts in the following compounds.
`CH_3(CH_2)_16COO(CH_2CH_2O)_nCH_2CH_2OH`
Define disinfectants with a Suitable example?
Which of the following is an example of liquid dishwashing detergent?
Polyethyleneglycols are used in the preparation of which type of detergents?
Which of the following statements are correct?
(i) Cationic detergents have germicidal properties.
(ii) Bacteria can degrade the detergents containing highly branched chains.
(iii) Some synthetic detergents can give foam even in ice cold water.
(iv) Synthetic detergents are not soaps.
Which category of the synthetic detergents is used in toothpaste?
Dishwashing soaps are synthetic detergents. What is their chemical nature?
Draw the diagram showing micelle formation by the following detergent.
\[\ce{CH3(CH2)10CH2OS\overset{-}{O}3\overset{+}{N}a}\]
Match structures given in Column I with the type of detergents given in Column II.
| Column I | Column II |
| (i) \[\ce{CH3(CH2)16COO(CH2CH2O) nCH2CH2OH}\] | (a) Cationic detergent |
| (ii) \[\ce{C17H35COO- Na+}\] | (b) Anionic detergent |
| (iii) \[\ce{CH3-(CH2)10CH2SO3- Na+}\] | (c) Nonionic detergent |
|
(iv) |
(d) Soap |
Synthetic detergents have advantage over usual soaps as far as cleansing power is concerned. But use of synthetic detergents over a long time creates environmental pollution. How can the pollution caused by synthetic detergents be minimised? Classify the detergents according to their chemical nature.
Explain the following term with suitable example.
Cationic detergents
Explain the following term with suitable examples.
cationic detergents
Explain the following terms with suitable examples Cationic detergents
Explain the following term with suitable example cationic detergents.
Explain the Following Term with Suitable Examples.
Cationic Detergents
Explain the following term with a suitable example:
Cationic detergents
Explain the following term with suitable example.
Cationic detergents
Explain the following term with suitable example:
cationic detergents