Advertisements
Advertisements
प्रश्न
What are biodegradable and non-biodegradable detergents? Give one example of each.
उत्तर १
Detergents that can be degraded by bacteria are called biodegradable detergents. Such detergents have straight hydrocarbon chains. For example: sodium lauryl sulphate
Detergents that cannot be degraded by bacteria are called non-biodegradable detergents. Such detergents have highly-branched hydrocarbon chains. For example: sodium -4- (1, 3, 5, 7- tetra methyl octyl) benzene sulphonate
उत्तर २
Detergents having straight chain hydrocarbons are easily degraded (or decomposed) by microorganisms and hence are called biodegradable detergents while detergents containing branched hydrocarbon chains are not easily degraded by the microorganisms find hence are called non-biodegradable detergents. Consequently, non-biodegradable detergents accumulate in rivers and water ways thereby causing severe water pollution. Examples of biodegradable detergents are sodium lauryl sulphate, sodium 4-(-l-dodecyl) benzenesulphonate and sodium 4-(2-dodecyl) benzenesulphonate.
Examples of non-biodegradable detergents is sodium 4-(1, 3,5,7 – tetramethyloctyl) benzene sulphonate.
APPEARS IN
संबंधित प्रश्न
Explain the following term with a suitable example:
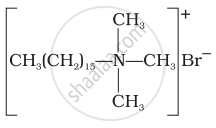
Cationic detergents
Explain the following terms with suitable examples - Anionic detergents
Explain the following terms with suitable examples - Non-ionic detergents
Label the hydrophilic and hydrophobic parts in the following compounds.
CH3(CH2)10CH2OSO3 –Na+
Label the hydrophilic and hydrophobic parts in the following compounds.
`CH_3(CH_2)_16COO(CH_2CH_2O)_nCH_2CH_2OH`
What are anionic detergents? Give an example ?
Define the following term with a suitable example:
Cationic detergents
What type of detergent are used in toothpastes?
Polyethyleneglycols are used in the preparation of which type of detergents?
Which of the following statements are correct?
(i) Cationic detergents have germicidal properties.
(ii) Bacteria can degrade the detergents containing highly branched chains.
(iii) Some synthetic detergents can give foam even in ice cold water.
(iv) Synthetic detergents are not soaps.
Which category of the synthetic detergents is used in toothpaste?
Hair shampoos belong to which class of synthetic detergent?
Draw the diagram showing micelle formation by the following detergent.
\[\ce{CH3(CH2)10CH2OS\overset{-}{O}3\overset{+}{N}a}\]
Match structures given in Column I with the type of detergents given in Column II.
| Column I | Column II |
| (i) \[\ce{CH3(CH2)16COO(CH2CH2O) nCH2CH2OH}\] | (a) Cationic detergent |
| (ii) \[\ce{C17H35COO- Na+}\] | (b) Anionic detergent |
| (iii) \[\ce{CH3-(CH2)10CH2SO3- Na+}\] | (c) Nonionic detergent |
|
(iv) |
(d) Soap |
Explain the following terms with suitable examples Cationic detergents
Explain the following term with a suitable example.
cationic detergents
Explain the following term with suitable examples.
Cationic detergents
Explain the following term with a suitable example:
Cationic detergents
Explain the following term with suitable example:
cationic detergents