Advertisements
Advertisements
प्रश्न
Why do soaps not work in hard water?
उत्तर
Soaps are sodium or potassium salts of long-chain fatty acids. Hard water contains calcium and magnesium ions. When soaps are dissolved in hard water, these ions displace sodium or potassium from their salts and form insoluble calcium or magnesium salts of fatty acids. These insoluble salts separate as scum.
`2C_17H_35COONa + CaCl -> 2NaCl + (C_17H_35COO)_2Ca`
Soap Insiluable calcium Stearate(Soap)
This is the reason why soaps do not work in hard water.
APPEARS IN
संबंधित प्रश्न
Why is bithional added to soap?
What is a soap ?
Write the chemical equation for preparing sodium soap from Glyceryl palmitate . Structural formulae of the compounds are given below.
(C15H31COO)3C3H5 – Glyceryl palmitate
Write the chemical equation for preparing sodium soap from glyceryl oleate . Structural formulae of the compounds are given below.
(C17H32COO)3C3H5 – Glyceryl oleate
Following type of nom-ionic detergents are present in liquid detergents, emulsifying agents and wetting agents. Label the hydrophilic and hydrophobic parts in the molecule. Identify the functional group (s) present in the molecule.
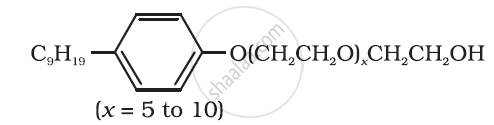
Explain the mechanism of cleansing action of soaps.
Which of the following enhances leathering property of soap?
Glycerol is added to soap. It functions ______.
What is a soft soap?
Why is it safer to use soap from the environmental point of view?
How are transparent soaps manufactured?
What are fillers and what role these fillers play in soap?
Match the soaps given in Column I with items given in Column II.
| Column I | Column II |
| (i) Soap chips | (a) dried miniature soap bubbles |
| (ii) Soap granules | (b) small broken pieces of soap formed from melted soaps |
| (iii) Soap powder | (c) soap powder + abrasives + builders \[\ce{(Na2CO3,Na3PO4)}\] |
| (iv) Scouring soap | (d) soap powder + builders like \[\ce{Na2CO3}\] and \[\ce{Na3PO4}\] |
Which of the following is not a correct statement?
Green chemistry in day-to-day life is in the use of ______.
