Advertisements
Advertisements
Question
Why do soaps not work in hard water?
Solution
Soaps are sodium or potassium salts of long-chain fatty acids. Hard water contains calcium and magnesium ions. When soaps are dissolved in hard water, these ions displace sodium or potassium from their salts and form insoluble calcium or magnesium salts of fatty acids. These insoluble salts separate as scum.
`2C_17H_35COONa + CaCl -> 2NaCl + (C_17H_35COO)_2Ca`
Soap Insiluable calcium Stearate(Soap)
This is the reason why soaps do not work in hard water.
APPEARS IN
RELATED QUESTIONS
Why is bithional added to soap?
What is a soap ?
Write the chemical equation for preparing sodium soap from Glyceryl palmitate . Structural formulae of the compounds are given below.
(C15H31COO)3C3H5 – Glyceryl palmitate
Following type of nom-ionic detergents are present in liquid detergents, emulsifying agents and wetting agents. Label the hydrophilic and hydrophobic parts in the molecule. Identify the functional group (s) present in the molecule.
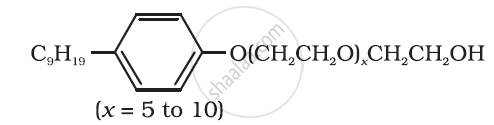
Explain the cleansing action of soaps.
Write balanced chemical equations for the action of hydrogen bromide on styrene in the presence of a peroxide
What is a soft soap?
If soap has high alkali content it irritates skin. How can the amount of excess alkali be determined? What can be the source of excess alkali?
Why is it safer to use soap from the environmental point of view?
What is the side product of soap industry? Give reactions showing soap formation.
What is the difference between bathing soap and washing soaps?
How are transparent soaps manufactured?
What are fillers and what role these fillers play in soap?
Match the detergents given in Column I with their uses given in Column II.
| Column I | Column II |
(i) 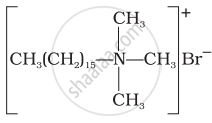 |
(a) Dishwashing powder |
(ii) 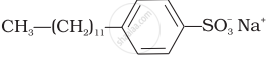 |
(b) Laundry soap |
| (iii) \[\ce{C17H33CO\overset{-}{O}\overset{+}{N}a + Na2CO3 + Rosin}\] | (c) Hair conditioners |
| (iv) \[\ce{CH3(CH2)16COO(CH2CH2O)nCH2CH2OH}\] | (d) Toothpaste |
Assertion: Transparent soaps are made by dissolving soaps in ethanol.
Reason: Ethanol makes things invisible.
Assertion: Sodium chloride is added to precipitate soap after saponification.
Reason: Hydrolysis of esters of long-chain fatty acids by alkali produces soap in colloidal form.
Which of the following is not a correct statement?
