Advertisements
Advertisements
प्रश्न
Explain the terms Tyndall effect.
उत्तर
Tyndall effect: - When a beam of light is allowed to pass through a colloidal solution, it becomes visible like a column of light. This is known as the Tyndall effect. This phenomenon takes place as particles of colloidal dimensions scatter light in all directions.
APPEARS IN
संबंधित प्रश्न
Out of BaCl2 and KCl, which one is more effective in causing coagulation of a negatively charged colloidal Sol? Give reason.
Explain what is observed When a beam of light is passed through a colloidal sol.
What happens when an emulsion is centrifuged?
Answer the following question.
Why are medicines more effective in the colloidal state?
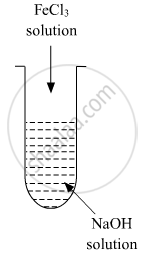
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the sol represented?
Which of the following will show Tyndall effect?
Which of the following statements are correct?
(i) Mixing two oppositely charged sols neutralises their charges and stabilises the colloid.
(ii) Presence of equal and similar charges on colloidal particles provides stability to the colloids.
(iii) Any amount of dispersed liquid can be added to emulsion without destabilising it.
(iv) Brownian movement stabilises sols.
Which phenomenon occurs when an electric field is applied to a colloidal solution and electrophoresis is prevented?
(i) Reverse osmosis takes place.
(ii) Electroosmosis takes place.
(iii) Dispersion medium begins to move.
(iv) Dispersion medium becomes stationary.
What causes brownian motion in colloidal dispersion?
Assertion: An ordinary filter paper impregnated with collodion solution stops the flow of colloidal particles.
Reason: Pore size of the filter paper becomes more than the size of colloidal particle.
Give reason for the following:
Lyophilic colloidal solutions are more stable than lyophobic colloidal solutions.
The important factor in the stabilization of a colloidal solution is ______
State Hardy-Schulze rule.
Define the following term:
Coagulation
In order to cause coagulation of 100 ml Arsenious sulphide sol 111.7 mg of 2 M NaCl solution is used, the coagulation value of NaCl will be ______.
6.84 g Al2(SO4)3 is needed to coagulate 2.5 L of As2S3 sol completely in 2.0 hrs. The coagulation value of Al2 (SO4)3 is ______.
