Advertisements
Advertisements
प्रश्न
Explain the term redox reaction with an example involving the reaction of hydrogen sulphide with chlorine.
उत्तर
Redox Reaction: “The reaction in which oxidation and reduction take place simultaneously is called Redox Reaction.”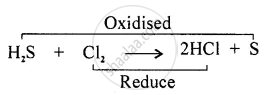
Removal of hydrogen is oxidation. Here `"H"_2"S"` is oxidised to sulphur whereas the addition of hydrogen is reduction.
Here chlorine is being reduced to hydrogen chloride.
APPEARS IN
संबंधित प्रश्न
Fill in the blank
Oxidation is the removal of ......................and addition of ....................
Identify from the following reaction the reactants that undergo oxidation and reduction.
2Ag2O → 4 Ag + O2 ↑
State, giving reason, whether the substances printed in bold letters have been oxidized or reduced.
PbO + CO → Pb + CO2
State, giving reason, whether the substances printed in bold letters have been oxidized or reduced.
Mg + 2HCl → MgCl2 + H2
State whether the following conversion is oxidation or reduction:
Cu2+ + 2 e-→ Cu
Which of the following statements about hydrogen is incorrect?
Name:
a liquid that is an oxidizing agent as well as a reducing agent.
Explain the term oxidation in term of addition and removal of oxygen/hydrogen with a suitable example.
A silver article generally turns black when kept in the open for a few days. The article when rubbed with toothpaste again starts shining.
- Why do silver articles turn black when kept in the open for a few days? Name the phenomenon involved.
- Name the black substance formed and give its chemical formula.
State the change in colour observed in following case mentioning the reason:
Copper powder is strongly heated in air.
