Advertisements
Advertisements
प्रश्न
Figure shows the P-V diagram of an ideal gas undergoing a change of state from A to B. Four different parts I, II, III and IV as shown in the figure may lead to the same change of state.

- Change in internal energy is same in IV and III cases, but not in I and II.
- Change in internal energy is same in all the four cases.
- Work done is maximum in case I
- Work done is minimum in case II.
उत्तर
b and c
Explanation:
Change in internal energy depends only upon the initial and final positions and not on the path followed. Hence, the change in internal energy is the same for all the cases. The work done is dependent upon the path followed. Work done is the area enclosed by the path with the x-axis. Since path I enclose the maximum area with the x-axis, the work done is maximum in case I.
APPEARS IN
संबंधित प्रश्न
Explain why Two bodies at different temperatures T1 and T2, if brought in thermal contact, do not necessarily settle to the mean temperature (T1 + T2)/2.
Explain why Air pressure in a car tyre increases during driving.
A gas is contained in a metallic cylinder fitted with a piston. The piston is suddenly moved in to compress the gas and is maintained at this position. As time passes the pressure of the gas in the cylinder ______________ .
What is the energy associated with the random, disordered motion of the molecules of a system called as?
One gram of water (1 cm3) becomes 1671 cm3 of steam at a pressure of 1 atm. The latent heat of vaporization at this pressure is 2256 J/g. Calculate the external work and the increase in internal energy.
A thermodynamic system goes from states
(i) P, V to 2P, V (ii) P, V to P, 2V
The work done in the two cases is ____________.
Which of the following represents isothermal process?
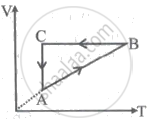
A cyclic process ABCA is shown in the V-T diagram. A process on the P-V diagram is ______.
A steam engine delivers 4.8 x 108 Jof work per minute and services 1.2 x 109 J of heat per minute from its boiler. What is the percentage efficiency of the engine?
A system releases 125 kJ of heat while 104 kJ work is done on the system. Calculate the change in internal energy.
