Advertisements
Advertisements
Question
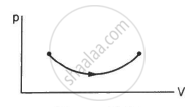
Figure shows the P-V diagram of an ideal gas undergoing a change of state from A to B. Four different parts I, II, III and IV as shown in the figure may lead to the same change of state.

- Change in internal energy is same in IV and III cases, but not in I and II.
- Change in internal energy is same in all the four cases.
- Work done is maximum in case I
- Work done is minimum in case II.
Solution
b and c
Explanation:
Change in internal energy depends only upon the initial and final positions and not on the path followed. Hence, the change in internal energy is the same for all the cases. The work done is dependent upon the path followed. Work done is the area enclosed by the path with the x-axis. Since path I enclose the maximum area with the x-axis, the work done is maximum in case I.
APPEARS IN
RELATED QUESTIONS
Explain why Air pressure in a car tyre increases during driving.
Should the internal energy of a system necessarily increase if heat is added to it?
An ideal gas is pumped into a rigid container having diathermic walls so that the temperature remains constant. In a certain time interval, the pressure in the container is doubled. Is the internal energy of the contents of the container also doubled in the interval ?
Consider the process on a system shown in figure. During the process, the work done by the system ______________ .
What is the energy associated with the random, disordered motion of the molecules of a system called as?
What is the internal energy of the system, when the amount of heat Q is added to the system and the system does not do any work during the process?
One gram of water (1 cm3) becomes 1671 cm3 of steam at a pressure of 1 atm. The latent heat of vaporization at this pressure is 2256 J/g. Calculate the external work and the increase in internal energy.
n mole of a perfect gas undergoes a cyclic process ABCA (see figure) consisting of the following processes:
A `→` B: Isothermal expansion at temperature T so that the volume is doubled from V1 to V2 = 2V1 and pressure changes from P1 to P2.
B `→` C: Isobaric compression at pressure P2 to initial volume V1.
C `→` A: Isochoric change leading to change of pressure from P2 to P1.
Total workdone in the complete cycle ABCA is ______.

If a gas is compressed adiabatically:
The internal energy of one mole of argon is ______.
