Advertisements
Advertisements
प्रश्न
Give its
(i) graphical representation,
(ii) mathematical expression
(iii) significance
उत्तर
(i) Graphical representation of Charles's law
T vs V: The relationship between the volume and the temperature of a gas can be plotted on a graph. A straight line is obtained.
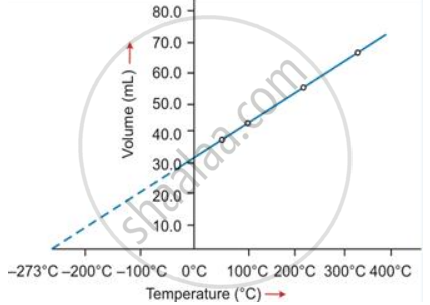
(ii) Mathematical expression
Let V0 be the volume of a fixed mass of a gas at 0 °C, and let V be its volume at temperature t °C at, constant pressure. then, according to Charles Law
`"V" = "V"_0 + "V"_0/273 t` (when P is constant)
`"V" = "V"_0 (1 + t/273) = "V"_0 ((273 + t)/273)` .......(1)
`"V" = "V"_0/273 "T" "where" "T" = 273 + "t"`
For a given mass of a gas,
`"V"_0/273 = "constant"`
`"V" = "k" xx "T"` (where k is constant)
(iii) Significance of Charles's Law: The volume of a given mass of a gas is directly proportional to its temperature; hence, the density decreases with temperature. This is the reason that
(a) Hot air is filled in balloons used for meteorological purposes. (b) Cable wires contract in winters and expand in summers.
APPEARS IN
संबंधित प्रश्न
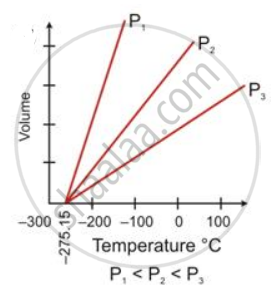
State the law which is represented by the following graph:
Give reasons for the following:
Mountaineers carry oxygen cylinders with them.
Choose the correct answer:
The volume-temperature relationship is given by
What will be the minimum pressure required to compress 500 dm3 of air at 1 bar to 200 dm3 temperature remaining constant?
88 cm3 of nitrogen is at a pressure of 770 mm mercury. If the pressure is raised to 880 mmHg, find by how much the volume will diminish, the temperature remaining constant.
Correct the following statement:
The volume of a fixed mass of a gas is directly proportional to its temperature, pressure remaining constant.
A certain mass of gas occupied 850 ml at a pressure of 760 mm of Hg. On increasing the pressure it was found that the volume of the gas was 75% of its initial value. Assuming constant temperature, find the final pressure of the gas?
It is required to reduce the volume of a gas by 20% by compressing it at a constant pressure. To do so, the gas has to be cooled. If the gas attains a final temperature of 157°C, find the initial temperature of the gas.
Fill in the blank with the correct word, from the words in option:
If the temperature of a fixed mass of a gas is kept constant and the pressure is increased, the volume correspondingly _______.
The pressure on one mole of gas at s.t.p. is doubled and the temperature is raised to 546 K. What is the final volume of the gas ? [one mole of a gas occupies a volume of 22.4 litres at stp.]
