Advertisements
Advertisements
प्रश्न
Give one example of an endothermic reaction.
उत्तर
Example of endothermic reaction: Barium hydroxide on reacting with ammonium chloride produces ammonia, water and barium chloride. It also results in decrease in temperature, as it consumes heat energy.
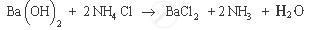
APPEARS IN
संबंधित प्रश्न
Balance the following equation:
Pb3O4 → PbO + O2
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate the mass of acid required.
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – mercury [II] oxide.
What is meant by ‘reactants’ and ‘products’ in a chemical equation?
Balance the following simple equation:
Al + H2O → Al2O3 + H2
Match the columns.
| Reactants | Products | Types of chemical reaction | ||
| 1. | MgH2 | → | Mg + H2 | Endothermic |
| 2. | 2H2S + SO2 | → | 3S + 2H2O | Oxidation |
| 3. | CaO + H2O | → | Ca(OH)2 + heat | Exothermic |
| Redox | ||||
Write three steps of writing chemical equations with example?
Balance the given chemical reaction as per the instructions below.
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Write the number of atoms of various elements in the reactants and products.
Reema took 5ml of Lead Nitrate solution in a beaker and added approximately 4ml of Potassium Iodide solution to it. What would she observe?
Three beakers labelled as A, B and C each containing 25 mL of water were taken. A small amount of NaOH, anhydrous CuSO4 and NaCl were added to the beakers A, B and C respectively. It was observed that there was an increase in the temperature of the solutions contained in beakers A and B, whereas in case of beaker C, the temperature of the solution falls. Which one of the following statement(s) is(are) correct?
- In beakers A and B, exothermic process has occurred.
- In beakers A and B, endothermic process has occurred.
- In beaker C exothermic process has occurred.
- In beaker C endothermic process has occurred.
