Advertisements
Advertisements
प्रश्न
Match the columns.
| Reactants | Products | Types of chemical reaction | ||
| 1. | MgH2 | → | Mg + H2 | Endothermic |
| 2. | 2H2S + SO2 | → | 3S + 2H2O | Oxidation |
| 3. | CaO + H2O | → | Ca(OH)2 + heat | Exothermic |
| Redox | ||||
उत्तर
| Reactants | Products | Types of chemical reaction | ||
| 1. | MgH2 | → | Mg + H2 | Oxidation |
| 2. | 2H2S + SO2 | → | 3S + 2H2O | Redox |
| 3. | CaO + H2O | → | Ca(OH)2 + heat | Exothermic |
APPEARS IN
संबंधित प्रश्न
Balance the following chemical equation.
\[\ce{BaCl2 + H2SO4 -> BaSO4 + HCl}\]
Translate the following statement into chemical equation and then balance it.
Hydrogen sulphide gas burns in the air to give water and sulphur dioxide.
Translate the following statement into chemical equation and then balance it.
Barium chloride reacts with aluminium sulphate to give aluminium chloride and a precipitate of barium sulphate.
Translate the following statement into chemical equation and then balance the equation:
Aluminium metal replaces iron from ferric oxide, Fe2O3, giving aluminium oxide and iron.
Correct and balance the following equation:
Ca + H2O  CaOH + H
CaOH + H
Balance the given chemical equation:
NH3 → N2 + H2
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are metals.
Aluminium burns in chlorine to form aluminium chloride (AlCl3). Write a balanced chemical equation for this reaction.
Convey the following information in the form of a balanced chemical equation:
"An aqueous solution of ferrous sulphate reacts with an aqueous solution of sodium hydroxide to form a precipitate of ferrous hydroxide and sodium sulphate remains in solution."
Write balanced chemical equation with state symbols for the following reaction:
Barium chloride solution reacts with sodium sulphate solution to give insoluble barium sulphate and a solution of sodium chloride.
Name the type of chemical reaction shown by the following equation:
Fe + CuSO4 → FeSO4 +Cu
Write word equation for the following skeletal equation:
\[\ce{CO + O2 -> CO2}\]
Balance the following chemical equation :
H2 + O2 → H2O
Write your observation for the following chemical reaction and name the product formed :
When dilute acetic acid is poured on baking soda.
Write symbolic representation for the following word equation and balance them :
Carbon + Oxygen → Carbon dioxide.
Balance the following equation. Also name the product formed.
`"Zn" + "HCI" → "ZnCI" _2 + "H"_2`
Write the chemical equation for the following word equation and balance them.
Calcium + Nitrogen → Calcium nitride
Write the chemical equation for the following word equation and balance them.
Sodium reacts with water to form sodium hydroxide and hydrogen
Balance the following equation:
Fe2O3 + CO → Fe + CO2
Write the balanced chemical equation of the following reaction.
potassium bicarbonate + sulphuric acid → potassium sulphate + carbon dioxide + water
Write the balanced chemical equation of the following reaction.
aluminium sulphate + sodium hydroxide → sodium sulphate + sodium meta aluminate + water.
Write the balanced chemical equation of the following reaction.
sodium chloride + manganese dioxide + sulphuric acid → sodium hydrogen sulphate + manganese sulphate + water + chlorine.
Name the substances that are getting oxidised and reduced in the process.
Write word equation for the following molecular equation:
\[\ce{CuSO4 + 2NaOH -> Na2SO4 + Cu(OH)2↓}\]
Word equation:
State the colour of the products.
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – mercury [II] oxide.
In certain reaction an insoluble solid called precipitate is formed. State the colour and name of the precipitate formed in the following reaction involving addition of:
Copper [II] sulphate to sodium hydroxide.
Give one example in the case where supplying energy [given below] is necessary for a chemical reaction.
Catalyst
Name the following:
Two gases which react under pressure in presence of a catalyst at elevated temperatures to give a gaseous product.
Write word equation for the following chemical reaction given below. Also state the observation seen in the case.
\[\ce{ZnCo3->[\triangle] ZnO + CO2}\]
Write word equation for the following chemical reaction given below. Also state the observation seen in the case.
\[\ce{Na2SO3 + 2HCl ->[\triangle] 2NaCl + H2O + SO2}\]
Balance the following simple equation:
CO2 + C → CO
Balance the following simple equation:
NaOH + Cl2 → NaCl + NaClO + H2O
Balance the following simple equation:
Fe2O3 + CO → Fe + CO2
Write a balanced equation for the following word equation:
Nitrogen dioxide + Water + Oxygen → Nitric acid
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
Na2SO3 + HCl → 2NaCl + H2O + SO2
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
Cl2 + 2H2O + SO2 → 4HCl + H2SO4
In the chemical equation the _______ are written on the left-hand side.
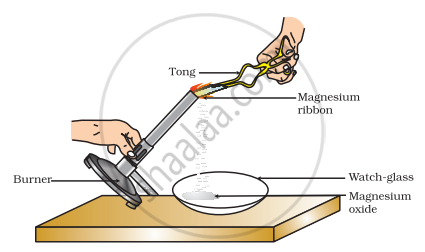
Which of the following is the correct observation of the reaction shown in the above set up?
