Advertisements
Advertisements
प्रश्न
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
उत्तर
The interparticle attraction is weak and interparticle space is large in gases because the particles are completely free to move randomly in the entire available space and take the shape of the vessel in which the gas is kept.
APPEARS IN
संबंधित प्रश्न
Molar volume is the volume occupied by 1 mol of any (ideal) gas at standard temperature and pressure (STP: 1 atmospheric pressure, 0 °C). Show that it is 22.4 litres
An oxygen cylinder of volume 30 litres has an initial gauge pressure of 15 atm and a temperature of 27 °C. After some oxygen is withdrawn from the cylinder, the gauge pressure drops to 11 atm and its temperature drops to 17 °C. Estimate the mass of oxygen taken out of the cylinder (R = 8.31 J mol–1 K–1, molecular mass of O2 = 32 u)
What do you understand by gas?
Correct the following statement:
0°C is equal to zero Kelvin.
If the absolute temperature of a body is doubled, the power radiated will increase by a factor of ______
Show that for monoatomic gas the ratio of the two specific heats is 5:3.
Estimate the average thermal energy of a helium atom at the temperature on the surface of the Sun (6000 K).
Which of the following diagrams (Figure) depicts ideal gas behaviour?
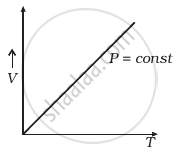 (a) |
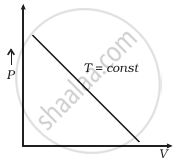 (b) |
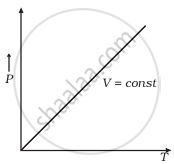 (c) |
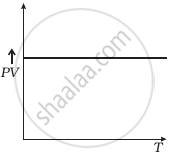 (d) |
Calculate the number of atoms in 39.4 g gold. Molar mass of gold is 197g mole–1.
P ∝ T at constant volume is the statement of ______.
