Advertisements
Advertisements
प्रश्न
Calculate the number of atoms in 39.4 g gold. Molar mass of gold is 197g mole–1.
उत्तर
We know that,
Molar mass = Mass of Avogadro's number of atoms (Molecules).
= 6.023 × 1023 atoms.
Given, molar mass of gold = 197 g/mol
Now,
∴ 197 g of gold contains = 6.023 × 1023 atoms
∴ 1 g of gold contain = `(6.023 xx 10^23)/197` atoms
∴ 39.4 g of gold contains = `(6.023 xx 10^23 xx 39.4)/197`
⇒ 1.20 × 1023 atoms.
APPEARS IN
संबंधित प्रश्न
The figure shows the plot of PV/T versus Pfor 1.00×10–3 kg of oxygen gas at two different temperatures.
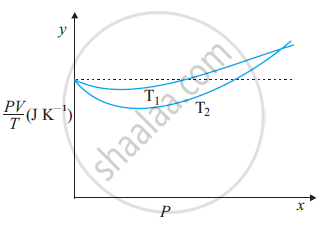
(a) What does the dotted plot signify?
(b) Which is true: T1 > T2 or T1 < T2?
(c) What is the value of PV/T where the curves meet on the y-axis?
(d) If we obtained similar plots for 1.00 ×10–3 kg of hydrogen, would we get the same value of PV/T at the point where the curves meet on the y-axis? If not, what mass of hydrogen yields the same value of PV/T (for low pressure high temperature region of the plot)? (Molecular mass of H2 = 2.02 u, of O2 = 32.0 u, R = 8.31 J mo1–1 K–1.)
Estimate the total number of air molecules (inclusive of oxygen, nitrogen, water vapour and other constituents) in a room of capacity 25.0 m3 at a temperature of 27 °C and 1 atm pressure
Estimate the average thermal energy of a helium atom at the temperature of 10 million Kelvin (the typical core temperature in the case of a star).
During the practical session in the lab when hydrogen sulphide gas having offensive odour is prepared for some test, we can smell the gas even 50 metres away. Explain the phenomenon.
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
Choose the correct answer:
The graph of PV vs P for gas is
A box of 1.00 m3 is filled with nitrogen at 1.50 atm at 300K. The box has a hole of an area 0.010 mm2. How much time is required for the pressure to reduce by 0.10 atm, if the pressure outside is 1 atm.
Cooking gas containers are kept in a lorry moving with uniform speed. The temperature of the gas molecules inside will ______.
P ∝ T at constant volume is the statement of ______.
Two tanks of equal volume contain equal mass of oxygen and nitrogen at 127°C. Find the ratio of pressure in two tanks.
