Advertisements
Advertisements
Question
Calculate the number of atoms in 39.4 g gold. Molar mass of gold is 197g mole–1.
Solution
We know that,
Molar mass = Mass of Avogadro's number of atoms (Molecules).
= 6.023 × 1023 atoms.
Given, molar mass of gold = 197 g/mol
Now,
∴ 197 g of gold contains = 6.023 × 1023 atoms
∴ 1 g of gold contain = `(6.023 xx 10^23)/197` atoms
∴ 39.4 g of gold contains = `(6.023 xx 10^23 xx 39.4)/197`
⇒ 1.20 × 1023 atoms.
APPEARS IN
RELATED QUESTIONS
What is diffusion? Give an example to illustrate it.
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
Fill in the blanks:
If the temperature is reduced to half, ………….. would also reduce to half.
Give reason for the following:
Volumes of gases are converted into s.t.p. conditions and then compared.
The average energy per molecule is proportional to ______
Show that for monoatomic gas the ratio of the two specific heats is 5:3.
Gases exert pressure on the walls of the container because the gas molecules ______
The equation of state for 2g of oxygen at a pressure 'P' and temperature 'T', when occupying a volume 'V' will be ______.
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Do the vessels contain an equal number of respective molecules?
Which of the following diagrams (Figure) depicts ideal gas behaviour?
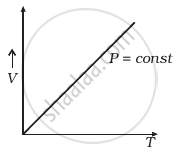 (a) |
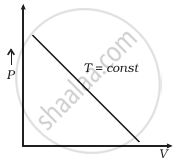 (b) |
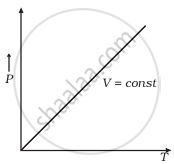 (c) |
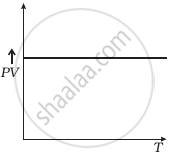 (d) |
