Advertisements
Advertisements
प्रश्न
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
2-Methylbutan-2-ol
उत्तर
\[\begin{array}{cc}
\ce{CH3}\phantom{............................}\ce{CH3}\phantom{................}\\
|\phantom{................................}|\phantom{..................}\\
\ce{CH3 - C - CH2CH3 + SOCl2 ->[\Delta]CH3 - C - CH2CH3 + SO2 + HCl}\\
|\phantom{................................}|\phantom{..................}\\
\ce{OH}\phantom{......................}\ce{\underset{2-chloro-2-methylbutane}{Cl}}\phantom{.........}\
\end{array}\]
संबंधित प्रश्न
Give two reactions that show the acidic nature of phenol.
Explain how does the −OH group attached to a carbon of benzene ring activate it towards electrophilic substitution?
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
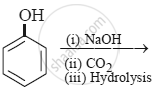
The product obtained from the reaction is:
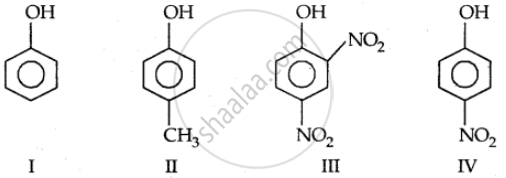
Strength of acidity is in order:
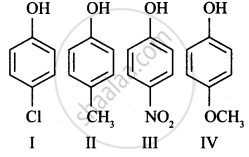
Arrange the following in decreasing order of acidic character:
Which of the following statements is true:
Mark the correct order of decreasing acid strength of the following compounds.
| (a) | 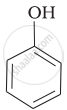 |
| (b) | 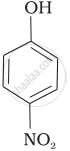 |
| (c) | 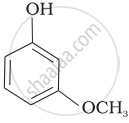 |
| (d) | 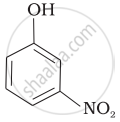 |
| (e) | 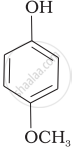 |
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
2-Methylbutan-2-ol
Give the structure of the product you would expect when the following alcohol reacts with HBr.
Butan-1-ol
