Advertisements
Advertisements
प्रश्न
In β-emission from a radioactive substance, an electron is ejected. This electron comes from ______.
विकल्प
the outermost orbit of an atom
the inner orbits of an atom
the surface of substance
the nucleus of an atom
उत्तर
In β-emission from a radioactive substance, an electron is ejected. This electron comes from the nucleus of an atom.
Explanation:
Radioactivity is a nuclear phenomenon. Hence, electrons come out of the nucleus. An electron is created as a result of the decay of one neutron into a proton inside the nucleus, and it is not possible for the electron to stay inside the nucleus; thus, it is spontaneously emitted.
APPEARS IN
संबंधित प्रश्न
What happens to the position of an element in the periodic table when its nucleus emits β -particle? Give reasons for your answer.
What kind of change takes place in a nucleus when a β - particle is emitted? Express it by an equation. State whether
- atomic number and
- mass number are conserved in a radioactive β - decay?
A nucleus is \[\ce {^24_11 Na} \] β-radioactive.
What general name is given to the product nucleus with respect to \[\ce{^24_11 Na}\]?
One roentgen is equal to _______ disintegrations per second.
Define critical mass.
A radioactive nucleus containing 128 nucleons emits a β-particle. After β- emission the number of nucleons present in the nucleus will be ______.
|
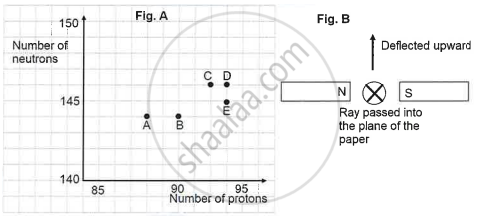
The graph (fig A) illustrates the correlation between the number of protons (x-axis) and the number of neutrons (y-axis) for elements A, B, C, D, and E in the periodic table. These elements are denoted by the letters rather than their conventional symbols. When the element C, depicted in the graph, undergoes radioactive decay, it releases radioactive rays. When these rays are directed into the plane of the paper in the presence of a magnetic field, as indicated in the fig B, they experience deflection, causing them to move upwards.
|
Name the radioactive radiations emitted by the element C.
|
The graph (fig A) illustrates the correlation between the number of protons (x-axis) and the number of neutrons (y-axis) for elements A, B, C, D, and E in the periodic table. These elements are denoted by the letters rather than their conventional symbols. When the element C, depicted in the graph, undergoes radioactive decay, it releases radioactive rays. When these rays are directed into the plane of the paper in the presence of a magnetic field, as indicated in the fig B, they experience deflection, causing them to move upwards.
|
Identify the daughter element from the graph.
What changes occur in the nucleus of a radioactive element when it emits a beta particle. Give one example, in support of your answer.
A nucleus \[\ce{^24_11Na}\] is β-radioactive.
Write the equation representing β-decay.