Advertisements
Advertisements
प्रश्न
In the two tetrahedral structures of dichromate ion, ______.
विकल्प
4 Cr–O bonds are equivalent in length
6 Cr–O bonds are equivalent in length
All Cr–O bonds are equivalent in length
All Cr–O bonds are non-equivalent
उत्तर
In the two tetrahedral structures of dichromate ion, 6 Cr–O bonds are equivalent in length.
Explanation:
The Dichromate di-anion is made up of two tetrahedral molecules that share oxygen at their common corner. The structure of the chromate ion is tetrahedral. Due to resonance, the six terminal bonds in the dichromate ion have the same bond length. As a result, all six Cr–O bonds are equivalent.
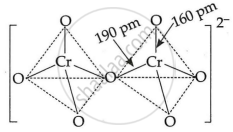
6 Cr–O bonds are equivalent
APPEARS IN
संबंधित प्रश्न
Complete the following equation:
\[\ce{2MnO4- + 6H+ + 5NO2- ->}\]
When chromite ore FeCr2O4 is fused with NaOH in presence of air, a yellow-coloured compound (A) is obtained, which on acidification with dilute sulphuric acid gives a compound (B). Compound (B) on reaction with KCl forms an orange coloured crystalline compound (C).
(i) Write the formulae of the compounds (A), (B) and C.
(ii) Write one use of compound (C).
Generally transition elements and their salts are coloured due to the presence of unpaired electrons in metal ions. Which of the following compounds are coloured?
(i) \[\ce{KMnO4}\]
(ii) \[\ce{Ce(SO4)2}\]
(iii) \[\ce{TiCl}\]
(iv) \[\ce{Cu2 Cl2}\]
When orange solution containing \[\ce{Cr2O^{2-}7}\] ion is treated with an alkali, a yellow solution is formed and when \[\ce{H^+}\] ions are added to yellow solution, an orange solution is obtained. Explain why does this happen?
Potassium permanganate acts as an oxidant in neutral, alkaline as well as acidic media. The final products obtained from it in the three conditions are, respectively.
Why \[\ce{HCl}\] should not be used for potassium permanganate titrations?
Complete the reaction mentioning all the products formed:
\[\ce{Cr2O^{2-}7 + 3H2S + 8H^+ ->}\]
Indicate the steps in the preparation of K2Cr2O7 from chromite ore.
Describe the preparation of potassium permanganate. How does the acidified permanganate solution react with SO2? Write the ionic equation for the reaction.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
