Advertisements
Advertisements
प्रश्न
In the two tetrahedral structures of dichromate ion, ______.
पर्याय
4 Cr–O bonds are equivalent in length
6 Cr–O bonds are equivalent in length
All Cr–O bonds are equivalent in length
All Cr–O bonds are non-equivalent
उत्तर
In the two tetrahedral structures of dichromate ion, 6 Cr–O bonds are equivalent in length.
Explanation:
The Dichromate di-anion is made up of two tetrahedral molecules that share oxygen at their common corner. The structure of the chromate ion is tetrahedral. Due to resonance, the six terminal bonds in the dichromate ion have the same bond length. As a result, all six Cr–O bonds are equivalent.
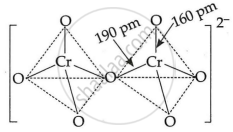
6 Cr–O bonds are equivalent
APPEARS IN
संबंधित प्रश्न
Complete the following equation:
\[\ce{2MnO4- + 6H+ + 5NO2- ->}\]
Complete the following chemical equation
Cr2O72- + 3Sn2+ + 14H+ →
Indicate the steps in the preparation of KMnO4 from pyrolusite ore.
Which of the following reactions are disproportionation reactions?
(a) \[\ce{Cu^{+} -> Cu^{2+} + Cu}\]
(b) \[\ce{3MnO^{-}4 + 4H^{+} -> 2MnO^{-}4 + MnO2 + 2H2O}\]
(c) \[\ce{2KMnO4 -> K2MnO4 + MnO2 + O2}\]
(d) \[\ce{2MnO^{-}4 + 3Mn^{2+} + 2H2O -> 5MnO2 + 4H^{+}}\]
Which of the following is amphoteric oxide?
\[\ce{Mn2O7, CrO3, Cr2O3, CrO, V2O5, V2O4}\]
KMnO4 is coloured due to ______.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Describe the preparation of potassium permanganate. How does the acidified permanganate solution react with SO2? Write the ionic equation for the reaction.
