Advertisements
Advertisements
प्रश्न
Which property of transition metals enables them to behave as catalysts?
पर्याय
High melting point
High ionisation enthalpy
Alloy formation
Variable oxidation states
उत्तर
Variable oxidation states
Explanation:
Because transition metals have partially full d-orbitals, they can easily extract electrons from or donate electrons to reagents, depending on the nature of the reaction.
They also have a high number of oxidation states and the potential to form complexes, making them excellent catalysts.
APPEARS IN
संबंधित प्रश्न
Which of the following cations are coloured in aqueous solutions and why ?
Sc3+, V3+, Ti4+, Mn2+ (At. Nos. Sc = 21, V = 23, Ti = 22, Mn = 25)
Complete the following chemical equations:
`(i) Cr_2O_7^(2-)+6Fe^(2+)+14H^+ ->`
`(ii) 2CrO_4^(2-)+2H^+ ->`
`(iii) 2MnO_4^-+5C_2O_4^(2-)+16H^+ ->`
Account for the following:
Cu+2 salts are coloured, while Zn2+ salts are white.
Give reasons for the following:
The transition metals generally form coloured compounds.
A solution of \[\ce{KMnO4}\] on reduction yields either a colourless solution or a brown precipitate or a green solution depending on pH of the solution. What different stages of the reduction do these represent and how are they carried out?
The standard electrode potentials of four elements A, B, C and D are – 3.05, – 1.66, – 0.40 and + 0.80. The highest chemical reactivity will be exhibited by
The trend of which property is represented by the following graph?
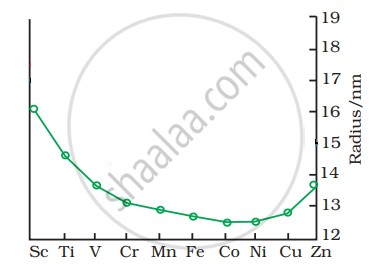
Explain the magnetic properties of d-block (or transition) elements.
Explain the use of different transition metals as catalysts.
Give a reason for the following:
Transition metals possess a great tendency to form complex compounds.
