Advertisements
Advertisements
Question
Which property of transition metals enables them to behave as catalysts?
Options
High melting point
High ionisation enthalpy
Alloy formation
Variable oxidation states
Solution
Variable oxidation states
Explanation:
Because transition metals have partially full d-orbitals, they can easily extract electrons from or donate electrons to reagents, depending on the nature of the reaction.
They also have a high number of oxidation states and the potential to form complexes, making them excellent catalysts.
APPEARS IN
RELATED QUESTIONS
ln which pair highest oxidation states of transition metals are found:
Out of Mn3+ and Cr3+, which is more paramagnetic and why ?
(Atomic nos. : Mn = 25, Cr = 24)
Explain why:
(i) Mn2+ is more stable than Fe2+ towards oxidation to +3 state.
(At. no. of Mn = 25, Fe = 26)
(ii) Transition elements usually form coloured ions.
(iii) Zr and Hf exhibit similar properties.
(At. no. of Zr = 40, Hf = 72)
Electronic configuration of a transition element X in +3 oxidation state is [Ar]3d5. What is its atomic number?
\[\ce{KMnO4}\] acts as an oxidising agent in acidic medium. The number of moles of \[\ce{KMnO4}\] that will be needed to react with one mole of sulphide ions in acidic solution is ______.
Although fluorine is more electronegative than oxygen, but the ability of oxygen to stabilise higher oxidation states exceeds that of fluorine. Why?
The complex showing a spin-span magnetic moment of 2.82 B.M. is :-
On adding NaOH, solution to the aqueous solution of K2CrO7 the colour of the solution changes from
The trend of which property is represented by the following graph?
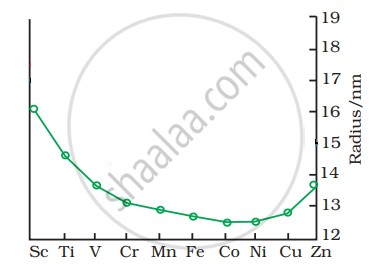
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following point:
Oxidation states
