Advertisements
Advertisements
Question
The trend of which property is represented by the following graph?
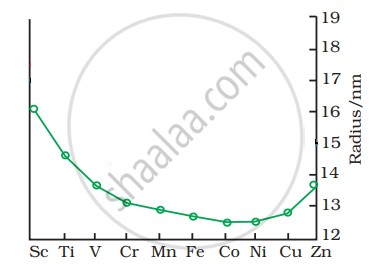
Options
ionization enthalpy
atomic radii
enthalpy of atomization
melting point
Solution
Atomic radii
APPEARS IN
RELATED QUESTIONS
Account for the following:
E° value for the Mn3+/Mn2+ couple is much more positive than that for Cr3+/Cr2+.
Explain briefly how +2 state becomes more and more stable in the first half of the first row transition elements with increasing atomic number?
Describe the oxidising action of potassium dichromate and write the ionic equation for its reaction with iodide.
The transition metals show _________ character because of the presence of unpaired· electrons and Cu+ is ____________ because of its electronic configuration is [Ar]3d10
Account for the following :
Ti4+ is colourless whereas V4+ is coloured in an aqueous solutions.
Electronic configuration of a transition element X in +3 oxidation state is [Ar]3d5. What is its atomic number?
Identify the metal and justify your answer.
\[\ce{MO3F}\]
The standard electrode potentials of four elements A, B, C and D are – 3.05, – 1.66, – 0.40 and + 0.80. The highest chemical reactivity will be exhibited by
Account for the following:
Sc3+ is colourless whereas Ti3+ is coloured in an aqueous solution.
Give two similarities in the properties of Sc and Zn.
