Advertisements
Advertisements
प्रश्न
Justify that the following reaction is redox reaction; identify the species oxidized/reduced, which acts as an oxidant and which acts as a reductant.
\[\ce{I2_{(aq)} + 2S2O^{2-}_{3(aq)}->S4O^{2-}_{6(aq)} + 2I^-_{ (aq)}}\]
उत्तर
\[\ce{I2_{(aq)} + 2S2O^{2-}_{3(aq)}->S4O^{2-}_{6(aq)} + 2I^-_{ (aq)}}\]
- Write oxidation number of all the atoms of reactants and products.
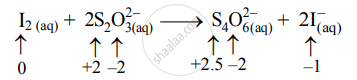
- Identify the species that undergoes a change in oxidation number.
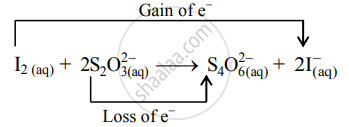
- The oxidation number of S increases from +2 to +2.5 and that of I decreases from 0 to –1. Because oxidation number of one species increases and that of the other decreases, the reaction is a redox reaction.
- The oxidation number of S increases by loss of electrons and therefore, S is a reducing agent and itself is oxidized. On the other hand, the oxidation number of I decreases by a gain of electrons, and therefore, I is an oxidizing agent and itself is reduced.
Result:
- The given reaction is a redox reaction.
- Oxidant/oxidizing agent (Reduced species): I2
- Reductant/reducing agent (Oxidized species): \[\ce{S2O^2-_3}\]
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
A compound contains atoms of three elements A, B, and C. If the oxidation state of A is +2, B is +5 and that of C is -2, the compound is possibly represented by
Choose the correct option.
The coefficients p, q, r, s in the reaction \[\ce{{p}Cr2O7^{2Θ} + {q}Fe^{2⊕}->{r}Cr^{3⊕} + {s}Fe^{3⊕} + H2O}\] respectively are:
Choose the correct option.
Oxidation number of carbon in H2CO3 is
Choose the correct option.
Which is the correct stock notation for manganese dioxide?
Calculate the oxidation number of the underlined atom.
HNO3
Identify the following pair of species is in its oxidized state.
Mg/Mg2+
Identify the following pair of species is in its oxidized state?
Cl2/Cl−
Provide the stock notation for the following compound:
Tl2O
Assign oxidation number atom in the following species.
\[\ce{Cr(OH)^Θ_4}\]
Assign oxidation number atom in the following species.
Na2S2O3
The following statements are CORRECT, EXCEPT:
What is the oxidation number of Mn in \[\ce{MnO^{2-}_4}\] ion?
In the reaction,
\[\ce{MnO^{-1}_4 (aq) + Br^{-1}(aq) -> MnO2(s) + BrO^{-1}_3(aq)}\]
the correct change in oxidation number of the species involved is ______.
Which of the following is NOT an example of redox reaction?
Identify the strongest oxidising agent.
\[\ce{Na^+ + e^- -> Na}\]; E0 = −2.714 V
\[\ce{Pt^{2+} + 2e^- -> Pt}\]; E0 = +1.200 V
\[\ce{I2 + 2e^- -> 2I^-}\]; E0 = + 0.535 V
\[\ce{Co^{2+} + 2e^- -> Co}\]; 0 = −0.280 V
Match the following.
| Compound | Oxidation no. of underlined element |
| i. \[\ce{\underline{C}_4H4O^{2-}_6}\] | a. +2.5 |
| ii. \[\ce{\underline{N}_3H}\] | b. +1.5 |
| iii. \[\ce{Mg2\underline{P}_2O7}\] | c. +5 |
| iv. \[\ce{Na2\underline{S}_4O6}\] | d. `-1//3` |
In which of the following, oxidation number of oxygen is +2?
Oxidation state of nitrogen in nitric oxide is ______.
The sum of oxidation states of all atoms in \[\ce{SnO^{2-}_2}\] and CO2 are ____________ respectively.
Which of the following is INCORRECT?
The oxidation state of phosphorous in Mg2P2O7 is ______.
The sum of oxidation number of all atoms in \[\ce{SnO^{2-}_3}\] ion is _______.
Which among the following pair of elements show highest oxidation state +7 in their different compounds?
Carbon is present in highest oxidation number in ______.
The oxidation number of phosphorous in Ba(H2PO2)2 is ______.
What is the oxidation state of chlorine atom in perchloric acid?
The oxidation number of oxygen in oxygen difluoride (OF2) and dioxygen difluoride (O2F2) respectively is ______.
