Advertisements
Advertisements
प्रश्न
Justify that the following reaction is redox reaction; identify the species oxidized/reduced, which acts as an oxidant and which acts as a reductant.
\[\ce{HF_{(aq)} + {OH}^-_{ (aq)}->H2O_{(l)} + {F}^ -_{ (aq)}}\]
उत्तर
\[\ce{HF_{(aq)} + {OH}^-_{ (aq)}->H2O_{(l)} + {F}^ -_{ (aq)}}\]
- Write oxidation number of all the atoms of reactants and products
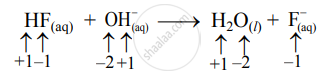
- Since the oxidation numbers of all the species remain the same, this is NOT a redox reaction.
Result:
The given reaction is NOT a redox reaction.
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
A compound contains atoms of three elements A, B, and C. If the oxidation state of A is +2, B is +5 and that of C is -2, the compound is possibly represented by
Choose the correct option.
Oxidation number of oxygen in superoxide is
Choose the correct option.
The process \[\ce{SO2->S2Cl2}\] is
Calculate the oxidation number of the underlined atom.
HNO3
Calculate the oxidation number of the underlined atom.
K2C2O4
Provide the stock notation for the following compound:
HAuCl4
Provide the stock notation for the following compound:
Tl2O
Provide the stock notation for the following compound:
FeO
Provide the stock notation for the following compound:
MnO
Provide the stock notation for the following compound:
CuO
Which of the following redox couple is a stronger oxidizing agent?
Cl2 (E0 = 1.36 V) and Br2 (E0 = 1.09 V)
Which of the following redox couple is a stronger reducing agent?
Zn (E0 = - 0.76 V) and Fe (E0 = - 0.44 V)
Oxidation state of Xe in XeOF4 is ____________.
The oxidation number of oxygen in peroxides is ____________.
What is the oxidation number of As in H3AsO3?
Which of the following is CORRECT?
\[\ce{H3PO4_{(aq)} + 3KOH_{(aq)} -> K3PO4_{(aq)} + 3H2O_{(l)}}\]
In the complex [Co(en)3]Cl3, ____________.
In which of the following, oxidation number of oxygen is +2?
In which among the following compounds, oxidation number of nitrogen is + 5?
Stock notations are used to specify the oxidation numbers of ____________.
The sum of oxidation number of all atoms in \[\ce{S2O^{2-}_3}\] ion is ______.
The sum of oxidation states of all atoms in \[\ce{Cr2O^{2-}_7}\] ion is ______.
The oxidation state of phosphorous in Mg2P2O7 is ______.
Carbon is present in highest oxidation number in ______.
The oxidation number of Cr in \[\ce{Cr(OH)^-_4}\] ion is ______.
The oxidation number of phosphorous in Ba(H2PO2)2 is ______.
What is the oxidation number of Cr in K2Cr2O7?
