Advertisements
Advertisements
प्रश्न
Methyl alcohol and water
- Give the principle involved in the separation of the mixture
- Give the technique of separation of the mixture.
उत्तर
By Fractional Distillation
Principle – Based on the separation of a mixture of two miscible liquids, with different boiling points.
Fractional distillation – It is the mixture of two miscible liquids with different boiling points is heated in a distillation flask when the two liquids separate out due to their difference in boiling points.
The technique of Separation – The mixture of two miscible liquids e.g. alcohol and water have different boiling points is taken in the distillation flask and heated slowly.
The mixture boils are the respective boiling points of the components and the component with the higher boiling point remains behind in the distillation flask condensation. The component with the lower boiling point collects in the receiver, after condensation in the Liebig's condenser.
The fractionating column contains several traps to allow the vapours of the higher boiling point components to condense, thereby only the lower boiling point component enters the condenser and is collected in the receiver.
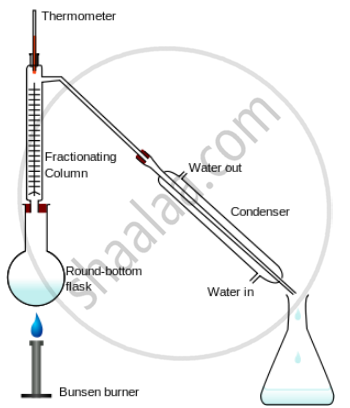
Fractionation distillation apparatus
APPEARS IN
संबंधित प्रश्न
How will you separate a mixture containing kerosene and petrol (difference in their boiling points is more than 25°C), which are miscible with each other?
What is the difference between distillation, distillation under reduced pressure and steam distillation ?
Define :- Distillation
State the correct technique for the separation of the following mixture.
a low boiling point liquid from a high boiling point liquid.
Choose the correct option.
Which of the following methods can be used to separate two compounds with different solubilities in the same solvent?
Choose the correct option.
Which of the following techniques is used for the separation of glycerol from soap in the soap industry?
What is the difference between simple distillation and fractional distillation?
Two liquids (A) and (B) can be separated by the method of fractional distillation. The boiling point of liquid (A) is less than boiling point of liquid (B). Which of the liquids do you expect to come out first in the distillate? Explain.
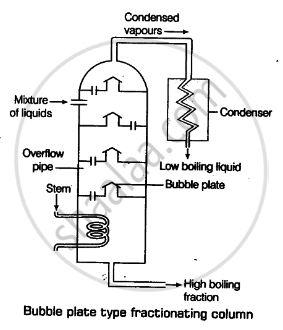
Draw a diagram of bubble plate type fractionating column. When do we require such type of a column for separating two liquids. Explain the principle involved in the separation of components of a mixture of liquids by using fractionating column. What industrial applications does this process have?
A mixture of benzene and chloroform is separated by ______.
