Advertisements
Advertisements
प्रश्न
Methyl alcohol and water
- Give the principle involved in the separation of the mixture
- Give the technique of separation of the mixture.
उत्तर
By Fractional Distillation
Principle – Based on the separation of a mixture of two miscible liquids, with different boiling points.
Fractional distillation – It is the mixture of two miscible liquids with different boiling points is heated in a distillation flask when the two liquids separate out due to their difference in boiling points.
The technique of Separation – The mixture of two miscible liquids e.g. alcohol and water have different boiling points is taken in the distillation flask and heated slowly.
The mixture boils are the respective boiling points of the components and the component with the higher boiling point remains behind in the distillation flask condensation. The component with the lower boiling point collects in the receiver, after condensation in the Liebig's condenser.
The fractionating column contains several traps to allow the vapours of the higher boiling point components to condense, thereby only the lower boiling point component enters the condenser and is collected in the receiver.
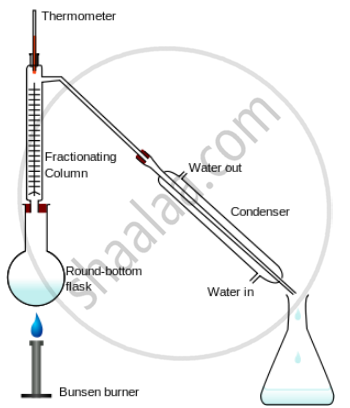
Fractionation distillation apparatus
APPEARS IN
संबंधित प्रश्न
How would you confirm that a colourless liquid given to you is pure water?
Give one word answer
The liquid which evaporates and then condenses during the process of distillation
Select the correct alternative from the choices given for the following statement:
Components of crude petroleum can be separated by
Give a scientific reason.
A wet cloth is wrapped around a water storage container in summer.
Pure water from impure water
- Give the principle involved in the separation of the mixture
- Give the technique of separation of the mixture.
Choose the correct option.
Which of the following techniques is used for the separation of glycerol from soap in the soap industry?
Match the type of mixture of compounds in Column I with the technique of separation/purification given in Column II.
| Column I | Column II |
| (i) Two solids which have different solubilities in a solvent and which do not undergo reaction when dissolved in it. | (a) Steam distillation |
| (ii) Liquid that decomposes at its boiling point | (b) Fractional distillation |
| (iii) Steam volatile liquid | (c) Simple distillation |
| (iv) Two liquids which have boiling points close to each other | (d) Distillation under reduced pressure |
| (v) Two liquids with large difference in boiling points. | (e) Crystallisation |
Assertion (A): Simple distillation can help in separating a mixture of propan-1-ol (boiling point 97°C) and propanone (boiling point 56°C).
Reason (R): Liquids with a difference of more than 20°C in their boiling points can be separated by simple distillation.
Two liquids (A) and (B) can be separated by the method of fractional distillation. The boiling point of liquid (A) is less than boiling point of liquid (B). Which of the liquids do you expect to come out first in the distillate? Explain.
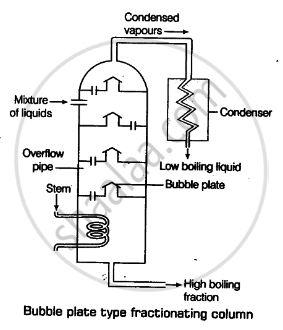
Draw a diagram of bubble plate type fractionating column. When do we require such type of a column for separating two liquids. Explain the principle involved in the separation of components of a mixture of liquids by using fractionating column. What industrial applications does this process have?
