Advertisements
Advertisements
Question
Methyl alcohol and water
- Give the principle involved in the separation of the mixture
- Give the technique of separation of the mixture.
Solution
By Fractional Distillation
Principle – Based on the separation of a mixture of two miscible liquids, with different boiling points.
Fractional distillation – It is the mixture of two miscible liquids with different boiling points is heated in a distillation flask when the two liquids separate out due to their difference in boiling points.
The technique of Separation – The mixture of two miscible liquids e.g. alcohol and water have different boiling points is taken in the distillation flask and heated slowly.
The mixture boils are the respective boiling points of the components and the component with the higher boiling point remains behind in the distillation flask condensation. The component with the lower boiling point collects in the receiver, after condensation in the Liebig's condenser.
The fractionating column contains several traps to allow the vapours of the higher boiling point components to condense, thereby only the lower boiling point component enters the condenser and is collected in the receiver.
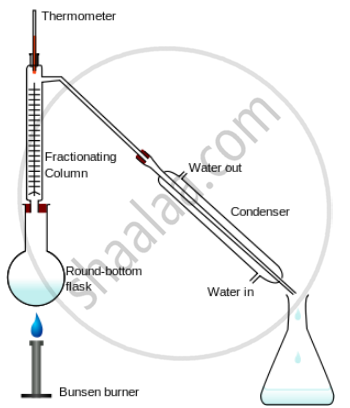
Fractionation distillation apparatus
APPEARS IN
RELATED QUESTIONS
What is the difference between distillation, distillation under reduced pressure and steam distillation ?
On what principle are the following methods of separation based? Give one example of a mixture for each of the methods mentioned in which they are used
Fractional distillation :
Explain the difference between Separation by distillation and by separating funnel.
Why is a condenser used in distillation process?
What will happen if the upper outlet of the condenser is connected to the tap instead of the lower outlet?
Alcohol can be separated from water by ______.
Which of the tubes in Fig. 2.1 (a) and (b) will be more effective as a condenser in the distillation apparatus?

Assertion (A): Simple distillation can help in separating a mixture of propan-1-ol (boiling point 97°C) and propanone (boiling point 56°C).
Reason (R): Liquids with a difference of more than 20°C in their boiling points can be separated by simple distillation.
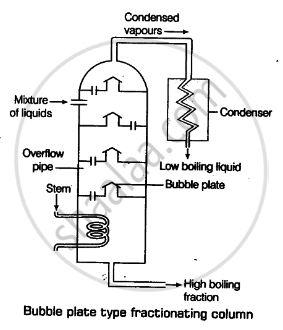
Draw a diagram of bubble plate type fractionating column. When do we require such type of a column for separating two liquids. Explain the principle involved in the separation of components of a mixture of liquids by using fractionating column. What industrial applications does this process have?
Explain the factors, which will affect the rate of evaporation, taking examples from our daily life experiences.
