Advertisements
Advertisements
प्रश्न
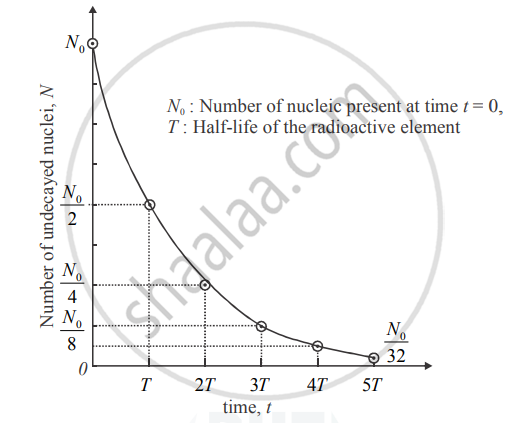
State the law of radioactive decay. hence derive the relation N = Noe-λt . Represent it graphically.
उत्तर
Radioactive decay law :
The number of nuclei undergoing the decay per unit time is proportional to the number of unchanged nuclei present at that instant.
Proof:
- Let ‘N’ be the number of nuclei present at any instant ‘t’.
- Let 'dN’ be the number of nucleic that disintegrated in short interval of time ‘dt’.
- According to law
`(-"dN")/"dt" ∝ "N"`
`therefore ("dN")/"dt"= -λ "N"`........................(1)
Where λ - decayconst
`therefore("dN")/"N"=-λ"dt"`
`therefore \text{integrating both sides}`
`∫ ("dN")/("N")=∫-λ "dt"`
` \[\ell\]"N"= λ t+C `................................(2)
Where C → integration consta
∴ at t = 0 , N =N0
∴ \[\ell\] nN0 = C .............(3)
`therefore ` Frome equation (2) and (3)
`l n N = -λ t +`\[\ell\] `nN_0`
`λ t = In\ N - In\ N_0`
-λT= In `(N/N_0)`
`N/N_0= e^-(λt)`
`therefore N=N_0e^-(λt)`
APPEARS IN
संबंधित प्रश्न
For the past some time, Aarti had been observing some erratic body movement, unsteadiness and lack of coordination in the activities of her sister Radha, who also used to complain of severe headache occasionally. Aarti suggested to her parents to get a medical check-up of Radha. The doctor thoroughly examined Radha and diagnosed that she has a brain tumour.
(a) What, according to you, are the values displayed by Aarti?
(b) How can radioisotopes help a doctor to diagnose brain tumour?
Write nuclear reaction equation for α-decay of `""_88^226"Ra"`.
Write nuclear reaction equation for α-decay of `""_94^242"Pu"`.
Write nuclear reaction equation for β+-decay of `""_6^11"C"`.
Write nuclear reaction equation for β+-decay of `""_43^97"Tc"`.
Write nuclear reaction equation for electron capture of `""_54^120"Xe"`.
Draw graphs showing variation of photoelectric current with applied voltage for two incident radiations of equal frequency and different intensities. Mark the graph for the radiation of higher intensity.
The sequence of stepwise decay of a radioactive nucleus is
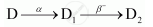
If the atomic number and mass number of D2 are 71 and 176 respectively, what are their corresponding values of D?
Radioactive 131I has a half-life of 8.0 days. A sample containing 131I has activity 20 µCi at t = 0. (a) What is its activity at t = 4 days? (b) What is its decay constant at t = 4.0 days?
The count rate from a radioactive sample falls from 4.0 × 106 per second to 1.0 × 106per second in 20 hours. What will be the count rate 100 hours after the beginning?
The half-life of 226Ra is 1602 y. Calculate the activity of 0.1 g of RaCl2 in which all the radium is in the form of 226Ra. Taken atomic weight of Ra to be 226 g mol−1 and that of Cl to be 35.5 g mol−1.
The selling rate of a radioactive isotope is decided by its activity. What will be the second-hand rate of a one month old 32P(t1/2 = 14.3 days) source if it was originally purchased for 800 rupees?
`""_80^197`Hg decay to `""_79^197`Au through electron capture with a decay constant of 0.257 per day. (a) What other particle or particles are emitted in the decay? (b) Assume that the electron is captured from the K shell. Use Moseley's law √v = a(Z − b) with a = 4.95 × 107s−1/2 and b = 1 to find the wavelength of the Kα X-ray emitted following the electron capture.
In an agricultural experiment, a solution containing 1 mole of a radioactive material (t1/2= 14.3 days) was injected into the roots of a plant. The plant was allowed 70 hours to settle down and then activity was measured in its fruit. If the activity measured was 1 µCi, what per cent of activity is transmitted from the root to the fruit in steady state?
4 × 1023 tritium atoms are contained in a vessel. The half-life of decay tritium nuclei is 12.3 y. Find (a) the activity of the sample, (b) the number of decay in the next 10 hours (c) the number of decays in the next 6.15 y.
238U decays to 206Pb with a half-life of 4.47 × 109 y. This happens in a number of steps. Can you justify a single half for this chain of processes? A sample of rock is found to contain 2.00 mg of 238U and 0.600 mg of 206Pb. Assuming that all the lead has come from uranium, find the life of the rock.
A human body excretes (removes by waste discharge, sweating, etc.) certain materials by a law similar to radioactivity. If technetium is injected in some form in a human body, the body excretes half the amount in 24 hours. A patient is given an injection containing 99Tc. This isotope is radioactive with a half-life of 6 hours. The activity from the body just after the injection is 6 μCi. How much time will elapse before the activity falls to 3 μCi?
In a gamma ray emission from nucleus :
Copy and complete the following table for a radioactive element whose half-life is 10 minutes. Assume that you have 30g of this element at t = 0.

Plot a graph showing the variation of undecayed nuclei N versus time t. From the graph, find out how one can determine the half-life and average life of the radioactive nuclei.
