Advertisements
Advertisements
प्रश्न
State the mass number, the atomic number, number of neutrons and electronic configuration of the following atoms.
`""^12""_6C, ""^16""_8O, ""^19""_9F, ""^20""_10Ne, ""_13""Al""^27, ""_17""Cl""^35`
Also, draw atomic diagrams for them.
उत्तर
| Name of elements | Atomic number | Atomic mass |
No of proton |
No of electrons |
no.of neutrons |
Electronic configuration |
| `""^12""_6C` | 6 | 12 | 6 | 6 | 6 | 2, 4 |
| `""^16""_8O` | 8 | 16 | 8 | 8 | 8 | 2, 6 |
| `""^19""_9F` | 9 | 19 | 9 | 9 | 10 | 2, 7 |
| `""^20""_10Ne` | 10 | 20 | 10 | 10 | 10 | 2, 8 |
| `""_13""Al""^27` | 13 | 27 | 13 | 13 | 14 | 2, 8, 3 |
| `""_17""Cl""^35` | 17 | 35 | 17 | 17 | 18 | 2, 8, 7 |
| Carbon `""^12""_6C` | 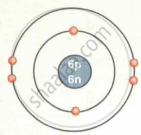 |
| Oxygen `""^16""_8O` | 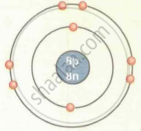 |
| Fluorine `""^19""_9F` | 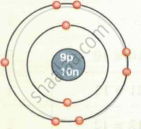 |
| Neon `""^20""_10Ne` | 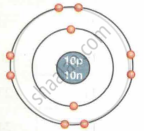 |
| Aluminium `""_13""Al""^27` | 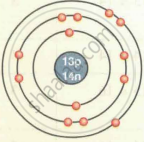 |
| Chlorine `""_17""Cl""^35` | 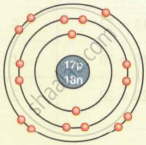 |
APPEARS IN
संबंधित प्रश्न
From the symbol `""_2^4"He"` for the element helium, write down the mass number and the atomic number of the element.
An atom of an element has two electrons in the M shell.
What is the (a) atomic number (b) number of protons in this element?
An element A atomic number 7 mass numbers 14
B electronic configuration 2,8,8
C electrons 13, neutrons 14
D Protons 18 neutrons 22
E Electronic configuration 2,8,8,1
State Valency of each element
An element A atomic number 7 mass numbers 14
B electronic configuration 2,8,8
C electrons 13, neutrons 14
D Protons 18 neutrons 22
E Electronic configuration 2,8,8,1
State (i) Valency of each element (ii) which one is a metal (iii) which is non-metal (iv) which is an inert gas
Define the term ‘mass number’ of an atom.
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Silicon [At. no. = 14, Mass no. = 28]
What is atomic number?
What is common salt? Name the elements present in it. Write the formula of common salt. What are the atomic number and the mass number of the elements? Write the ions in the compound.
What is a neutron?
When an atom gives electrons from its outermost shell, it becomes ______ charged.
Mass No. 27 is for the element ______.
