Advertisements
Advertisements
प्रश्न
State the mass number, the atomic number, number of neutrons and electronic configuration of the following atoms.
`""^12""_6C, ""^16""_8O, ""^19""_9F, ""^20""_10Ne, ""_13""Al""^27, ""_17""Cl""^35`
Also, draw atomic diagrams for them.
उत्तर
| Name of elements | Atomic number | Atomic mass |
No of proton |
No of electrons |
no.of neutrons |
Electronic configuration |
| `""^12""_6C` | 6 | 12 | 6 | 6 | 6 | 2, 4 |
| `""^16""_8O` | 8 | 16 | 8 | 8 | 8 | 2, 6 |
| `""^19""_9F` | 9 | 19 | 9 | 9 | 10 | 2, 7 |
| `""^20""_10Ne` | 10 | 20 | 10 | 10 | 10 | 2, 8 |
| `""_13""Al""^27` | 13 | 27 | 13 | 13 | 14 | 2, 8, 3 |
| `""_17""Cl""^35` | 17 | 35 | 17 | 17 | 18 | 2, 8, 7 |
| Carbon `""^12""_6C` | 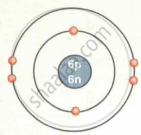 |
| Oxygen `""^16""_8O` | 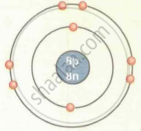 |
| Fluorine `""^19""_9F` | 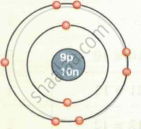 |
| Neon `""^20""_10Ne` | 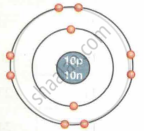 |
| Aluminium `""_13""Al""^27` | 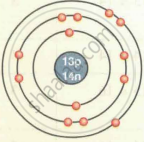 |
| Chlorine `""_17""Cl""^35` | 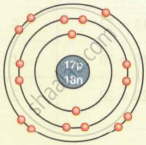 |
APPEARS IN
संबंधित प्रश्न
With the help of given Table, find out the mass number of oxygen.
| Composition of Atoms of the First Eighteen Elements with Electron Distribution in Various Shells | ||||||||||
| Name of Element | Symbol | Atomic Number | Number of protons | Number of Neutrons | Number of Electrons | Distribution of Electrons | Valency | |||
| K | L | M | N | |||||||
| Hydrogen | H | 1 | 1 | - | 1 | 1 | - | - | - | 1 |
| Helium | He | 2 | 2 | 2 | 2 | 2 | - | - | - | 0 |
| Lithium | Li | 3 | 3 | 4 | 3 | 2 | 1 | - | - | 1 |
| Beryllium | Be | 4 | 4 | 5 | 4 | 2 | 2 | - | - | 2 |
| Boron | B | 5 | 5 | 6 | 5 | 2 | 3 | - | - | 3 |
| Carbon | C | 6 | 6 | 6 | 6 | 2 | 4 | - | - | 4 |
| Nitrogen | N | 7 | 7 | 7 | 7 | 2 | 5 | - | - | 3 |
| Oxygen | O | 8 | 8 | 8 | 8 | 2 | 6 | - | - | 2 |
| Fluorine | F | 9 | 9 | 10 | 9 | 2 | 7 | - | - | 1 |
| Neon | Ne | 10 | 10 | 10 | 10 | 2 | 8 | - | - | 0 |
| Sodium | Na | 11 | 11 | 12 | 11 | 2 | 8 | 1 | - | 1 |
| Magnesium | Mg | 12 | 12 | 12 | 12 | 2 | 8 | 2 | - | 2 |
| Aluminium | Al | 13 | 13 | 14 | 13 | 2 | 8 | 3 | - | 3 |
| Silicon | Si | 14 | 14 | 14 | 14 | 2 | 8 | 4 | - | 4 |
| Phosphorus | P | 15 | 15 | 16 | 15 | 2 | 8 | 5 | - | 3.5 |
| Sulphur | S | 16 | 16 | 16 | 16 | 2 | 8 | 6 | - | 2 |
| Chlorine | Cl | 17 | 17 | 18 | 17 | 2 | 8 | 7 | - | 1 |
| Argon | Ar | 18 | 18 | 22 | 18 | 2 | 8 | 8 | 0 | |
Multiple Choice Questions
The sum of number of protons and number of neutrons present in the nucleus of an atom is called its
WRITE SHORT ANSWER
Define the atomic mass number of an atom.
State the main postulates of Dalton’s atomic theory.
If an atom ‘A’ has mass number 35 and atomic number 17, state the number of protons, electrons and neutrons it contains.
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Calcium – \[\ce{^40_20Ca}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
Match the statements in List I with the correct answer from List II.
| List I | List II |
| 1. Mass number of an atom is the number of protons and | A: Electron |
| 2. The sub-atomic particle with a negligible mass. | B: Argon |
| 3. An atom having stable electronic configuration. | C: Nitrogen |
| 4. A molecule formed by sharing of electrons [covalency] | D: Sodium |
| 5. A metallic atom having unstable electronic configuration. | E: Neutrons |
Differentiate between the following term:
Atomic number and mass number
The ratio of the radii of hydrogen atom and its nucleus is ~ 105. Assuming the atom and the nucleus to be spherical,
- what will be the ratio of their sizes?
- If atom is represented by planet earth ‘Re ’ = 6.4 ×106 m, estimate the size of the nucleus.
Explain the following terms.
- Proton
- Electron
- Neutron.
