Advertisements
Advertisements
Question
State the mass number, the atomic number, number of neutrons and electronic configuration of the following atoms.
`""^12""_6C, ""^16""_8O, ""^19""_9F, ""^20""_10Ne, ""_13""Al""^27, ""_17""Cl""^35`
Also, draw atomic diagrams for them.
Solution
| Name of elements | Atomic number | Atomic mass |
No of proton |
No of electrons |
no.of neutrons |
Electronic configuration |
| `""^12""_6C` | 6 | 12 | 6 | 6 | 6 | 2, 4 |
| `""^16""_8O` | 8 | 16 | 8 | 8 | 8 | 2, 6 |
| `""^19""_9F` | 9 | 19 | 9 | 9 | 10 | 2, 7 |
| `""^20""_10Ne` | 10 | 20 | 10 | 10 | 10 | 2, 8 |
| `""_13""Al""^27` | 13 | 27 | 13 | 13 | 14 | 2, 8, 3 |
| `""_17""Cl""^35` | 17 | 35 | 17 | 17 | 18 | 2, 8, 7 |
| Carbon `""^12""_6C` | 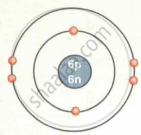 |
| Oxygen `""^16""_8O` | 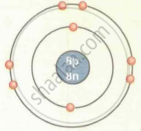 |
| Fluorine `""^19""_9F` | 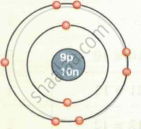 |
| Neon `""^20""_10Ne` | 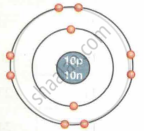 |
| Aluminium `""_13""Al""^27` | 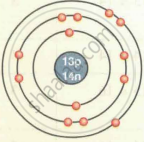 |
| Chlorine `""_17""Cl""^35` | 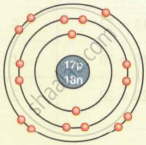 |
APPEARS IN
RELATED QUESTIONS
Fill in the blank of the following statement :
The total number of protons and neutrons in the nucleus of an atom is called its _________.
Fill in the following blanks :
| Atomic Number | Mass Number | Protons | Neutrons | Electrons | Symbol |
| 10 | 22 | _________ | _________ | _________ | _________ |
Give reason
Actual atomic mass is greater than mass number.
An element A atomic number 7 mass numbers 14
B electronic configuration 2,8,8
C electrons 13, neutrons 14
D Protons 18 neutrons 22
E Electronic configuration 2,8,8,1
State Valency of each element
An element A atomic number 7 mass numbers 14
B electronic configuration 2,8,8
C electrons 13, neutrons 14
D Protons 18 neutrons 22
E Electronic configuration 2,8,8,1
State (i) Valency of each element (ii) which one is a metal (iii) which is non-metal (iv) which is an inert gas
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Carbon [At. no. = 6, Mass no. = 12]
Explain the following terms.
- Proton
- Electron
- Neutron.
______ = No. of protons = No. of the electrons.
Mass No. 27 is for the element ______.
Ca2+ has completely filled outer shell. Justify your answer.
In which one of the following, the number of protons is greater than the number of neutrons but number of protons is less than the number of electrons?
