Advertisements
Advertisements
प्रश्न
Using crystal field theory, draw energy level diagram, write electronic configuration of the central metal atom/ion and determine the magnetic moment value in the following:
\[\ce{[FeF6]^{3-}, [Fe(H2O)6]^{2+}, [Fe(CN)6]^{4-}}\]
उत्तर
(1) \[\ce{FeF^{3-}6}\]:
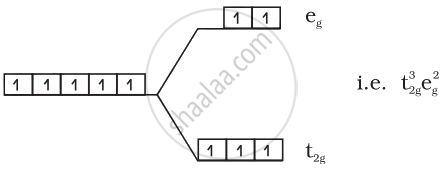
\[\ce{Fe^{3+} = 3d^5}\]
Number of unpaired electrons = 5
Magnetic moment = `sqrt(5(5 + 2))` = 5.92 BM
(2) \[\ce{[Fe(H2O)6]^{2+}}\]:
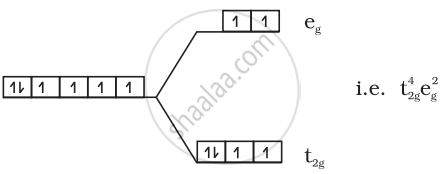
\[\ce{Fe^{2+} = 3d^6}\]
Number of unpaired electrons = 4
Magnetic moment = `sqrt(4(4 + 2))` = 4.9 BM
(3) \[\ce{[Fe(CN)6]^{4-}}\]:
\[\ce{Fe^{2+} = 3d^6}\]
Since CN– is a strong field ligand all the electrons get paired.

No unpaired electrons so diamagnetic.
APPEARS IN
संबंधित प्रश्न
Write the electronic configuration of Fe(III) on the basis of crystal field theory when it forms an octahedral complex in the presence of (i) strong field, and (ii) weak field ligand. (Atomic no.of Fe=26)
On the basis of crystal field theory explain why Co(III) forms paramagnetic octahedral complex with weak field ligands whereas it forms diamagnetic octahedral complex with strong field ligands.
Why are low spin tetrahedral complexes not formed?
Match the complex ions given in Column I with the hybridisation and number of unpaired electrons given in Column II and assign the correct code:
| Column I (Complex ion) | Column II (Hybridisation, number of unpaired electrons) |
| A. \[\ce{[Cr(H2O)6]^{3+}}\] | 1. dsp2, 1 |
| B. \[\ce{[Co(CN)4]^{2-}}\] | 2. sp3d2, 5 |
| C. \[\ce{[Ni(NH3)6]^{2+}}\] | 3. d2sp3, 3 |
| D. \[\ce{[MnF6]^{4-}}\] | 4. sp3, 4 |
| 5. sp3d2, 2 |
Using crystal field theory, draw energy level diagram, write electronic configuration of the central metal atom/ion and determine the magnetic moment value in the following:
\[\ce{[CoF6]^{3-}, [Co(H2O)6]^{2+}, [Co(Cn)6]^{3-}}\]
The correct order of increasing crystal field strength in following series:
Crystal field stabilising energy for high spind4 octahedral complex is:-
The magnitude of CFSE depends upon ______
Consider that d6 metal ion (M2+) forms a complex with aqua ligands and the spin only magnetic moment of the complex is 4.90 BM. The geometry and the crystal field stabilization energy of the complex is:
On the basis of Crystal Field Theory, write the electronic configuration of d4 ion if Δ0 > P.
