Advertisements
Advertisements
प्रश्न
Write the electronic configuration of Fe(III) on the basis of crystal field theory when it forms an octahedral complex in the presence of (i) strong field, and (ii) weak field ligand. (Atomic no.of Fe=26)
उत्तर
`"Fe"= ["Ar"]3"d"^6 4"s"^2`
`"Fe"^{3+} = ["Ar"]3"d"^6 4"s"^2`
`"Fe"^(+3) = ["AR"] 3"d"^5 4"s"^0`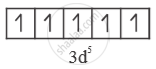
In octahedral crystal field.
(i) Strong field ligand - Electrons will be paired.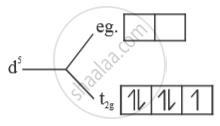
No. of unpaired e- = 1
Paramagnetic
`μ = sqrt(n(n+2) = sqrt(1(3) = sqrt3 B.M`
(ii) Weak field ligand
- Electrons follow Hund’s rule 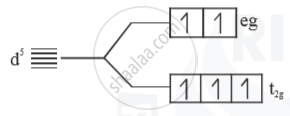
No. of unpaired = 5
Paramagnetic
`μ =sqrt(5(5+2))= sqrt(35) B.M.`
APPEARS IN
संबंधित प्रश्न
On the basis of crystal field theory, write the electronic configuration for d4 ion if Δ0 > P.
On the basis of crystal field theory, write the electronic configuration for d4 ion if ∆0 < P.
How are the following conversions carried out?
Benzoic acid into metanitrobenzoic acid.
Draw the structures of the following :
(1) XeF6
(2) IF7
Atomic number of \[\ce{Mn}\], \[\ce{Fe}\] and \[\ce{Co}\] are 25, 26 and 27 respectively. Which of the following inner orbital octahedral complex ions are diamagnetic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Mn(CN)6]^{3-}}\]
(iii) \[\ce{[Fe(CN)6]^{4-}}\]
(iv) \[\ce{[Fe(CN)6]^{3-}}\]
Atomic number of \[\ce{Mn, Fe, Co}\] and Ni are 25, 26, 27 and 28 respectively. Which of the following outer orbital octahedral complexes have same number of unpaired electrons?
(i) \[\ce{[MnCl6]^{3-}}\]
(ii) \[\ce{[FeF6]^{3-}}\]
(iii) \[\ce{[CoF6]^{3-}}\]
(iv) \[\ce{[Ni(NH3)6]^{2+}}\]
[Ni(H2O)6]2+ (aq) is green in colour whereas [Ni(H2O)4 (en)]2+ (aq)is blue in colour, give reason in support of your answer.
Considering crystal field theory, strong-field ligands such as CN–:
What is the spectrochemical series?
On the basis of Crystal Field Theory, write the electronic configuration of d4 ion if Δ0 > P.
