Advertisements
Advertisements
प्रश्न
We would like to make a vessel whose volume does not change with temperature (take a hint from the problem above). We can use brass and iron `(β_(vbrass) = (6 xx 10^(–5))/K and β_(viron) = (3.55 xx 10^(–5))/K)` to create a volume of 100 cc. How do you think you can achieve this.
उत्तर
In the previous problem, the difference in the length was constant.
In this problem the difference in volume is constant.
The situation is shown in the diagram.
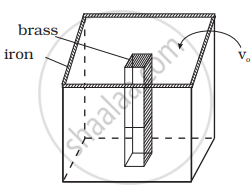
Let Vio, Vbo be the volume of iron and brass vessel at 0°C
Vi, Vb be the volume of iron and brass vessel at Δθ°C,
γi, γb be the coefficient of volume expansion of iron and brass.
As per the question, Vio – Vbo = 100 cc = Vi – Vb ......(i)
Now, `V_i = V_(io) (1 + γ_iΔθ)`
`V_b = V_(bo) (1 + γ_bΔθ)`
`V_i - V_b = (V_(io) - V_(bo)) + Δθ(V_(io)γ_i - V_(bo)γ_b)`
Since, `V_i - V_b` = constant
So, `V_(io)γ_i - V_(bo)γ_b`
⇒ `V_(io)/V_(bo) = γ_b/γ_i`
= `(3/2 β_b)/(3/2 β_i)`
= `β_b/β_i`
= `(6 xx 10^-5)/(3.55 xx 10^-5)`
= `6/3.55`
`V_(io)/V_(bo) = 6/3.55` ......(ii)
Solving equations (i) and (ii), we get
Vio = 244.9 cc
Vbo = 144.9 cc
APPEARS IN
संबंधित प्रश्न
Water in lakes and ponds do not freeze at once in cold countries. Give a reason is support of your answer.
Heat supplied to a solid change it into liquid. What is this change in the phase called?
Differentiate between heat capacity and specific heat capacity.
Write the expression for the heat energy Q received by the substance when m kg of substance of specific heat capacity c Jkg-1 k-1 is heated through Δt° C.
In an experiment to determine the specific heat capacity of a solid following operations were
made:
Mass of calorimeter + stirrer = x kg
Mass of water = y kg
Initial temperature of water t1℃
Mass of solid = z kg
Temperature of solid = t2 ℃
Temperature of mixture = t ℃
Specific heat capacity of calorimeter and water are c1 and c2 respectively. Express the specific
heat capacity c of the solid in terms of the above data.
It is generally cold after a hail-storm then during and before the hail storm. Give reason.
What impact will climate changes have on the crops of food?
What is carbon tax?
Solve the following problems:
Equal heat is given to two objects A and B of mass 1 g. Temperature of A increases by 3°C and B by 5°C. Which object has more specific heat? And by what factor?
The product of mass and specific heat is known as ..........
If 10125 J of heat energy boils off 4.5 g of water at 100°C to steam at 100°C, find the specific latent heat of steam.
The farmers fill their fields with water in winter. Give reason.
Explain, why do sandy soils, get heated up quickly as compared to wet soils?
A certain amount of heat Q will warm 1 g of material X by 3°C and 1 g of material Y by 4°C. Which material has a higher specific heat capacity?
Describe a method to determine the specific heat capacity of a solid (say, a piece of copper).
How much heat energy is necessary to raise the temperature of 5 kg of water from 20°C to 100°C?
Explain why the specific heat capacity at constant pressure is greater than the specific heat capacity at constant volume.
Express the change in internal energy in terms of molar specific heat capacity.
Which of the substances P, Q, or R has the lowest specific heat? The temperature v/s time graph is shown ______.

