Advertisements
Advertisements
प्रश्न
We would like to make a vessel whose volume does not change with temperature (take a hint from the problem above). We can use brass and iron `(β_(vbrass) = (6 xx 10^(–5))/K and β_(viron) = (3.55 xx 10^(–5))/K)` to create a volume of 100 cc. How do you think you can achieve this.
उत्तर
In the previous problem, the difference in the length was constant.
In this problem the difference in volume is constant.
The situation is shown in the diagram.
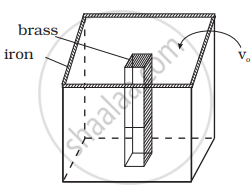
Let Vio, Vbo be the volume of iron and brass vessel at 0°C
Vi, Vb be the volume of iron and brass vessel at Δθ°C,
γi, γb be the coefficient of volume expansion of iron and brass.
As per the question, Vio – Vbo = 100 cc = Vi – Vb ......(i)
Now, `V_i = V_(io) (1 + γ_iΔθ)`
`V_b = V_(bo) (1 + γ_bΔθ)`
`V_i - V_b = (V_(io) - V_(bo)) + Δθ(V_(io)γ_i - V_(bo)γ_b)`
Since, `V_i - V_b` = constant
So, `V_(io)γ_i - V_(bo)γ_b`
⇒ `V_(io)/V_(bo) = γ_b/γ_i`
= `(3/2 β_b)/(3/2 β_i)`
= `β_b/β_i`
= `(6 xx 10^-5)/(3.55 xx 10^-5)`
= `6/3.55`
`V_(io)/V_(bo) = 6/3.55` ......(ii)
Solving equations (i) and (ii), we get
Vio = 244.9 cc
Vbo = 144.9 cc
APPEARS IN
संबंधित प्रश्न
What property of water makes it an effective coolant?
Heat supplied to a solid change it into liquid. What is this change in the phase called?
A heater of power P watt raises the temperature of m kg of a liquid by Δt K in time t s. Express
the specific heat capacity of liquid in terms of above data.
Name two green house gases ?
What impact will global warming have on the health of the affected population?
Give one example where high specific heat capacity of water is used as cooling.
A piece of iron of mass 2.0 kg has a heat capacity of 966 J K-1. Find its specific heat capacity in S.I unit.
What is heat? What is the S. I. unit of heat?
Explain, why does a wise farmer water his fields, if forecast is forst?
Ice-cream at 0°C feels colder than water at 0°C. Give reason for this observation.
1 kg of water freezes to form ice at 0°C. What amount of heat is withdrawn?
An electric immersion heater is rated 1250 W. Calculate the time in which it will heat 20 kg of water at 5°C to 65°C.
The SI unit of specific heat is _______.
Decide the unit for specific heat capacity.
Numerical Problem.
What is the heat in joules required to raise the temperature of 25 grams of water from 0°C to 100°C? What is the heat in Calories? (Specific heat of water = `(4.18"J")/("g"°"C")`
Numerical Problem.
What could be the final temperature of a mixture of 100 g of water at 90 °C and 600g of water at 20°C.
Heat is applied to a rigid diatomic gas at constant pressure. The ratio ΔQ : ΔU : ΔW is ______.
A piece of lead weighing 500 g gives out 1200 calories of heat when it is cooled from 100° C to 20° C. Find its specific heat.
On supplying 100 µC of charge to a conductor, its potential rises by 5 V then capacity of the conductor is ______.
When two kilocalories of heat are supplied to a system, the internal energy of the system increases by 5030 J and the work done by the gas against the external pressure is 3350 J. Calculate J, the mechanical equivalent of heat.
