Advertisements
Advertisements
Question
We would like to make a vessel whose volume does not change with temperature (take a hint from the problem above). We can use brass and iron `(β_(vbrass) = (6 xx 10^(–5))/K and β_(viron) = (3.55 xx 10^(–5))/K)` to create a volume of 100 cc. How do you think you can achieve this.
Solution
In the previous problem, the difference in the length was constant.
In this problem the difference in volume is constant.
The situation is shown in the diagram.
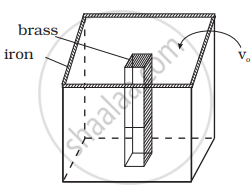
Let Vio, Vbo be the volume of iron and brass vessel at 0°C
Vi, Vb be the volume of iron and brass vessel at Δθ°C,
γi, γb be the coefficient of volume expansion of iron and brass.
As per the question, Vio – Vbo = 100 cc = Vi – Vb ......(i)
Now, `V_i = V_(io) (1 + γ_iΔθ)`
`V_b = V_(bo) (1 + γ_bΔθ)`
`V_i - V_b = (V_(io) - V_(bo)) + Δθ(V_(io)γ_i - V_(bo)γ_b)`
Since, `V_i - V_b` = constant
So, `V_(io)γ_i - V_(bo)γ_b`
⇒ `V_(io)/V_(bo) = γ_b/γ_i`
= `(3/2 β_b)/(3/2 β_i)`
= `β_b/β_i`
= `(6 xx 10^-5)/(3.55 xx 10^-5)`
= `6/3.55`
`V_(io)/V_(bo) = 6/3.55` ......(ii)
Solving equations (i) and (ii), we get
Vio = 244.9 cc
Vbo = 144.9 cc
APPEARS IN
RELATED QUESTIONS
A liquid X has the maximum specific heat capacity and is used as a coolant in Car Radiators. Name the liquid X.
During the phase change does the average kinetic energy of the molecules of the substance increase?
Water is used in hot water bottles for fomentation. Give a reason.
Give one example where high specific heat capacity of water is used as heat reservoir ?
State the impact of global warming on life on the earth.
What impact will global warming have on the health of the affected population?
Who shall pay carbon tax ?
What are the factors on which the quantity of heat given to a body depends?
Write the approximate values of the specific latent heat of fusion of ice.
Explain, why is water sprayed on roads in evening in hot summer?
Water falls from a height of 50 m. Calculate the rise in the temperature of water when it strikes the bottom.
(g = 10 ms-2; Specific heat capacity of water = 4200 J / kg°C)
How much heat energy is released when 5 g of water at 20° C changes to ice at 0° C?
[Specific heat capacity of water = 4.2 J g-1 ° C-1 Specific latent heat of fusion of ice = 336 J g-1]
Calculate the amount of heat released when 5.0 g of water at 20°C is changed into ice at 0°C.
(Specific heat capacity of water = 4.2 J/g°C
Specific latent heat of fusion of ice = 336 J/g)
Which of the following substances (A, B and C) has the highest specific beat?

The molar specific heats of an ideal gas at constant pressure and constant volume are denoted by Cp and Cv respectively. If `gamma = "C"_"p"/"C"_"v"` and R is the universal gas constant, then Cp is equal to ______.
Two cylinders of equal height and radius are made of copper and aluminum. Which of them conducts heat faster?
When two kilocalories of heat are supplied to a system, the internal energy of the system increases by 5030 J and the work done by the gas against the external pressure is 3350 J. Calculate J, the mechanical equivalent of heat.
