Advertisements
Advertisements
प्रश्न
What are the limitations of Rutherford’s model of the atom?
उत्तर
(i) An electron orbiting around the nucleus is accelerated towards it. An accelerating charged particle must produce radiation and lose energy. Thus, electrons in an atom must constantly emit radiation and lose energy. Because of this energy loss, the electron will slow down and cannot withstand the nucleus' attraction. As a result, the electron should spiral and eventually fall into the nucleus (see image).
If this occurs, the atom should collapse in approximately 10-8 seconds. However, this does not occur; atoms are stable. This suggests that something is amiss with Rutherford's nuclear model of atoms.
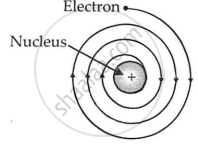
(ii) Rutherford's atomic model says nothing about how electrons are arranged in an atom.
APPEARS IN
संबंधित प्रश्न
On the basis of Rutherford’s model of an atom, which subatomic particle is present in the nucleus of an atom?
In Rutherford’s experiment, generally the thin foil of heavy atoms, like gold, platinum etc. have been used to be bombarded by the α-particles. If the thin foil of light atoms like aluminium etc. is used, what difference would be observed from the above results?
Give the evidence for the existence of nucleus in an atom.
How was it shown that an atom has a lot of empty space within it?
Name the particles used by Rutherford in his experiment on the discovery of nucleus. Also state the charge on these particles.
Rutherford's alpha particle scattering experiment led to the discovery of :
Draw a neat labelled diagram
Rutherford's scattering experiment
Proton was discovered by ______.
Which of the following conclusions could not be derived from Rutherford’s α -particle scattering experiment?
In Rutherford’s experiment, the bouncing of alpha particles suggests that the nucleus of an atom is negatively charged.
