Advertisements
Advertisements
प्रश्न
What is the effect of adding a catalyst on Activation energy (Ea)
उत्तर
The catalyst provides an alternate pathway or reaction mechanism by reducing the activation energy (Ea) between reactants and products.
APPEARS IN
संबंधित प्रश्न
The activation energy for the reaction \[\ce{2 HI_{(g)} -> H2_{(g)} + I2_{(g)}}\] is 209.5 kJ mol−1 at 581K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
The rate of chemical reaction becomes double for every 10° rise in temperature because of ____________.
Activation energy of a chemical reaction can be determined by ______.
Consider figure and mark the correct option.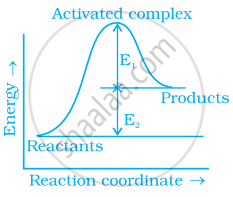
Mark the incorrect statements:
(i) Catalyst provides an alternative pathway to reaction mechanism.
(ii) Catalyst raises the activation energy.
(iii) Catalyst lowers the activation energy.
(iv) Catalyst alters enthalpy change of the reaction.
Match the statements given in Column I and Column II
| Column I | Column I | |
| (i) | Catalyst alters the rate of reaction | (a) cannot be fraction or zero |
| (ii) | Molecularity | (b) proper orientation is not there always |
| (iii) | Second half life of first order reaction | (c) by lowering the activation energy |
| (iv) | `e^((-E_a)/(RT)` | (d) is same as the first |
| (v) | Energetically favourable reactions (e) total probability is one are sometimes slow | (e) total probability is one |
| (vi) | Area under the Maxwell Boltzman curve is constant | (f) refers to the fraction of molecules with energy equal to or greater than activation energy |
Total number of vibrational degrees of freedom present in CO2 molecule is
The rate constant for a reaction is 1.5 × 10–7 sec–1 at 50°C. What is the value of activation energy?
The activation energy in a chemical reaction is defined as ______.
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below
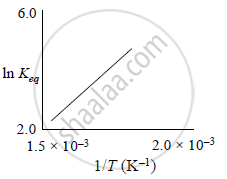
The reaction must be:
