Advertisements
Advertisements
प्रश्न
Match the statements given in Column I and Column II
| Column I | Column I | |
| (i) | Catalyst alters the rate of reaction | (a) cannot be fraction or zero |
| (ii) | Molecularity | (b) proper orientation is not there always |
| (iii) | Second half life of first order reaction | (c) by lowering the activation energy |
| (iv) | `e^((-E_a)/(RT)` | (d) is same as the first |
| (v) | Energetically favourable reactions (e) total probability is one are sometimes slow | (e) total probability is one |
| (vi) | Area under the Maxwell Boltzman curve is constant | (f) refers to the fraction of molecules with energy equal to or greater than activation energy |
उत्तर
| Column I | Column I | |
| (i) | Catalyst alters the rate of reaction | (c) by lowering the activation energy |
| (ii) | Molecularity | (a) cannot be fraction or zero |
| (iii) | Second half life of first order reaction | (d) is same as the first |
| (iv) | `e^((-E_a)/(RT)` | (f) refers to the fraction of molecules with energy equal to or greater than activation energy |
| (v) | Energetically favourable reactions (e) total probability is one are sometimes slow | (b) proper orientation is not there always |
| (vi) | Area under the Maxwell Boltzman curve is constant | (e) total probability is one |
Explanation:
(i) Catalyst alters the rate of reaction by lowering activation energy.
(ii) Molecularity cannot be fraction or zero. If molecularity is zero, reaction is not possible.
(iii) Second half-life of first order reaction is same as first because half-life
time is temperature independent. ‘
(iv) `e^((-E_a)/(RT)` refers to the fraction of molecules with kinetic energy equal to greater than activation energy.
(v) Energetically favourble reactions are sometimes slow due to improper orientation of molecule cause some ineffective collision of molecules.
(vi) Area under the Maxwell-Boltzmann curve is constant because total probability of molecule taking part in a chemical reaction is equal to one.
APPEARS IN
संबंधित प्रश्न
(b) Rate constant ‘k’ of a reaction varies with temperature ‘T’ according to the equation:
`logk=logA-E_a/2.303R(1/T)`
Where Ea is the activation energy. When a graph is plotted for `logk Vs. 1/T` a straight line with a slope of −4250 K is obtained. Calculate ‘Ea’ for the reaction.(R = 8.314 JK−1 mol−1)
The rate constant for the first-order decomposition of H2O2 is given by the following equation:
`logk=14.2-(1.0xx10^4)/TK`
Calculate Ea for this reaction and rate constant k if its half-life period be 200 minutes.
(Given: R = 8.314 JK–1 mol–1)
The rate of the chemical reaction doubles for an increase of 10 K in absolute temperature from 298 K. Calculate Ea.
The activation energy for the reaction \[\ce{2 HI_{(g)} -> H2_{(g)} + I2_{(g)}}\] is 209.5 kJ mol−1 at 581K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
The rate constant for the decomposition of hydrocarbons is 2.418 × 10−5 s−1 at 546 K. If the energy of activation is 179.9 kJ/mol, what will be the value of pre-exponential factor?
Write a condition under which a bimolecular reaction is kinetically first order. Give an example of such a reaction. (Given : log2 = 0.3010,log 3 = 0.4771, log5 = 0.6990).
Why does the rate of a reaction increase with rise in temperature?
Why in the redox titration of \[\ce{KMnO4}\] vs oxalic acid, we heat oxalic acid solution before starting the titration?
An exothermic reaction X → Y has an activation energy 30 kJ mol-1. If energy change ΔE during the reaction is - 20 kJ, then the activation energy for the reverse reaction in kJ is ______.
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below
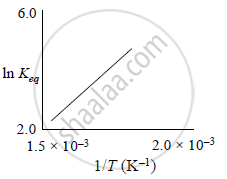
The reaction must be:
