Advertisements
Advertisements
प्रश्न
(b) Rate constant ‘k’ of a reaction varies with temperature ‘T’ according to the equation:
`logk=logA-E_a/2.303R(1/T)`
Where Ea is the activation energy. When a graph is plotted for `logk Vs. 1/T` a straight line with a slope of −4250 K is obtained. Calculate ‘Ea’ for the reaction.(R = 8.314 JK−1 mol−1)
उत्तर
`log k=logA-E_a/2.303R(1/T)`
Ea → Activation energy
The above equation is like y = mx + c where if we plot y v/s x we get a straight line with slope ‘m’ and intercept ‘c’.
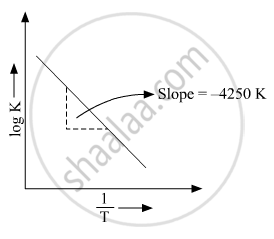
So, slope is equal to`=-E_a/2.303R`
`-E_a/2.303R=-4250k=>E_a=4250 xx 2.303 xx 8.314 =81,375.3535 j mol^(-1)`
`E_a=81.3753 KJ mol^(-1)`
APPEARS IN
संबंधित प्रश्न
The decomposition of A into product has value of k as 4.5 × 103 s−1 at 10°C and energy of activation 60 kJ mol−1. At what temperature would k be 1.5 × 104 s−1?
Define activation energy.
A first-order reaction is 50% completed in 40 minutes at 300 K and in 20 minutes at 320 K. Calculate the activation energy of the reaction. (Given : log 2 = 0·3010, log 4 = 0·6021, R = 8·314 JK–1 mol–1)
The decomposition of a hydrocarbon has value of rate constant as 2.5×104s-1 At 27° what temperature would rate constant be 7.5×104 × 3 s-1if energy of activation is 19.147 × 103 J mol-1 ?
The rate of chemical reaction becomes double for every 10° rise in temperature because of ____________.
Why does the rate of a reaction increase with rise in temperature?
In respect of the eqn k = \[\ce{Ae^{{-E_a}/{RT}}}\] in chemical kinetics, which one of the following statement is correct?
The activation energy in a chemical reaction is defined as ______.
The equation k = `(6.5 xx 10^12 "s"^(-1))"e"^(- 26000 " K"//"T")` is followed for the decomposition of compound A. The activation energy for the reaction is ______ kJ mol-1. (Nearest integer) (Given: R = 8.314 JK-1 mol-1)
An exothermic reaction X → Y has an activation energy 30 kJ mol-1. If energy change ΔE during the reaction is - 20 kJ, then the activation energy for the reverse reaction in kJ is ______.
