Advertisements
Advertisements
प्रश्न
What is lanthanoid contraction?
उत्तर १
The atomic and ionic radii of lanthanoids gradually decrease with an increase in atomic number. This process is known as lanthanoid contraction.
उत्तर २
In the lanthanoid series, as the atomic number increases, the atomic and ionic radii decrease from one element to the next, but this decrease is very small. For example – on going from Ce to Lu the atomic radius decreases from 183 pm to 173 pm and this decrease is only 10 pm. Similarly, on going from Ce3+ to Lu3+ ion the ionic radius decreases from 103 pm to 85 pm, which is only 18 pm. Hence, for an increase of 14 in atomic number, the decrease in atomic and ionic radii is much less. This decrease is much less than the elements of other groups and periods.
Table - Variation in atomic and ionic radii of lanthanum and lanthanoids (pm)
| Element | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu |
| Radius (Ln) | 187 | 183 | 182 | 181 | 181 | 180 | 199 | 180 | 178 | 177 | 176 | 175 | 174 | 173 | - |
| Radius (Ln3+) | 106 | 103 | 101 | 99 | 98 | 96 | 95 | 94 | 92 | 91 | 89 | 88 | 87 | 85 | - |
The steady decrease in atomic and ionic sizes of lanthanoid elements with an increase in atomic number is called 'lanthanoid contraction'.
The decrease in ionic radii of trivalent lanthanoids (Ln3+) is shown in.
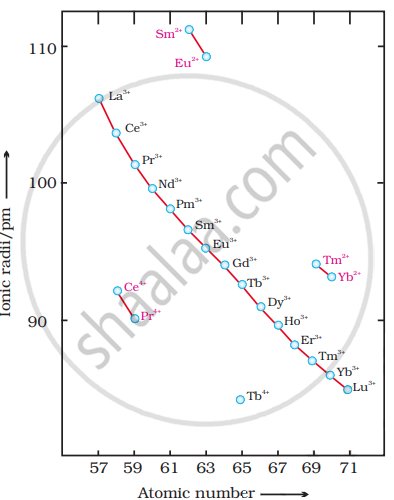
Causes of Lanthanide Contraction: In the lanthanoid series, on going from one element to the next, the nuclear charge increases by one unit and an electron is added. These new electrons are added to the parallel 4f-subshells. However, the shielding effect of one 4f-electron on the other 4f-electron (from the nuclear charge) is less due to the very large size of the f-orbitals. Although the nuclear charge increases by one unit at each step, the effective nuclear charge experienced by each 4f-electron increases with the increase in atomic number and nuclear charge. As a result, the entire 4f-electron shell contracts with the addition of each element, although this reduction is minimal. As a result, a regular decrease in the size of lanthanoids is observed with increasing atomic number. The sum of successive reductions gives the total lanthanoid contraction.
Notes
Students can refer to the provided solutions based on their preferred marks.
APPEARS IN
संबंधित प्रश्न
What are chemical twins? Write ‘two’ examples.
What are lanthanoids?
Account for the following :
Zr and Hf have almost similar atomic radii.
Name a member of the lanthanoid series which is well known to exhibit +4 oxidation state.
Compare the chemistry of actinoids with that of the lanthanoids with special reference to oxidation state.
Compare the chemistry of actinoids with that of the lanthanoids with special reference to atomic and ionic sizes.
Compare the chemistry of actinoids with that of the lanthanoids with special reference to chemical reactivity.
Write the electronic configurations of the elements with the atomic numbers 61, 91, 101 and 109.
Write the different oxidation states of iron
Explain, why lanthanum (Z = 57) forms La3+ ion, while cerium (Z = 58) forms Ce4+ ion?
What are the consequences of lanthanoid contraction?
Explain the cause of lanthanoid contraction?
Answer the followiiig questions:
Which trivalent ion has maximum size in the Lanthanoid series i.e. Lanthanum ion (La3+) to Luteium ion (Lu3+)?
(at. no. of Lanthanum = 57 and Lutetium = 71)
What is lanthanoid contraction? Write the.............
Which of the following lanthanoids show +2 oxidation state besides the characteristic oxidation state +3 of lanthanoids?
(i) \[\ce{Ce}\]
(ii) \[\ce{Eu}\]
(iii) \[\ce{Yb}\]
(iv) \[\ce{Ho}\]
Although Zr belongs to 4d and Hf belongs to 5d transition series but it is quite difficult to separate them. Why?
Although +3 oxidation states is the characteristic oxidation state of lanthanoids but cerium shows +4 oxidation state also. Why?
Match the compounds/elements given in Column I with uses given in Column II.
| Column I (Compound/element) | Column II (Use) | |
| (i) | Lanthanoid oxide | (a) Production of iron alloy |
| (ii) | Lanthanoid | (b) Television screen |
| (iii) | Misch metal | (c) Petroleum cracking |
| (iv) | Magnesium based alloy is constituent of | (d) Lanthanoid metal + iron |
| (v) | Mixed oxides of lanthanoids are employed | (e) Bullets |
| (f) In X-ray screen |
Match the statements given in Column I with the oxidation states given in Column II.
| Column I | Column II | |
| (i) | Oxidation state of Mn in MnO2 is | (a) + 2 |
| (ii) | Most stable oxidation state of Mn is | (b) + 3 |
| (iii) | Most stable oxidation state of | (c) + 4 |
| Mn in oxides is | (d) + 5 | |
| (iv) | Characteristic oxidation state of lanthanoids is | (e) + 7 |
Match the property given in Column I with the element given in Column II.
| Column I (Property) | Column II (Element) | |
| (i) | Lanthanoid which shows +4 oxidation state |
(a) Pm |
| (ii) | Lanthanoid which can show +2 oxidation state |
(b) Ce |
| (iii) | Radioactive lanthanoid | (c) Lu |
| (iv) | Lanthanoid which has 4f7 electronic configuration in +3 oxidation state |
(d) Eu |
| (v) | Lanthanoid which has 4f14 electronic configuration in +3 oxidation state |
(e) Gd |
| (f) Dy |
On the basis of Lanthanoid contraction, explain the following:
Nature of bonding in \[\ce{La2O3}\] and \[\ce{Lu2O3}\] .
On the basis of Lanthanoid contraction, explain the following:
Trends in the stability of oxo salts of lanthanoids from \[\ce{La}\] to \[\ce{Lu}\].
On the basis of Lanthanoid contraction, explain the following:
Stability of the complexes of lanthanoids.
How would you account for the following:
There is a greater range of oxidation states among the actinoids than among the lanthanides.
In lanthanoid the last electron enters (n - 2)f subshell where n is equal to ______.
Which one of the lanthanoids given below is the most stable in divalent form?
Mischmetal is an alloy consisting mainly of ______.
Write a note on lanthanoids.
State a reason for the following:
La(OH)3 is more basic than Lu(OH)3.
Why is Mn2+ ion more stable than Fe2+ ion?
(Atomic numbers of Mn = 25 and Fe = 26)
Assertion: There is a continuous increase in size among Lanthanoids with an increase in atomic number.
Reason: Lanthanoids do not show Lanthanoid contraction.
