Advertisements
Advertisements
प्रश्न
What is the effect of threshold frequency and stopping potential on increasing the frequency of the incident beam of light? Justify your answer.
उत्तर
Threshold frequency: The threshold frequency is the lowest frequency of incident radiation that can cause an electron to be ejected from a metal. At frequencies below the threshold, there is no photoelectric emission.
The frequency of light that will produce an emission of electrons from the metal's surface is referred to as the threshold frequency.
If ν signifies the frequency of the incident photon and νth signifies threshold frequency, then;
- If ν < νth, then this denotes that no ejection of photoelectrons will occur.
- If ν = νth, then this denotes that photoelectrons are just ejected from the surface of the metal, however, the kinetic energy of the electron is equal to zero.
The minimal negative voltage that must be provided to the anode to halt the photocurrent is known as stopping potential. When expressed in electron volts, the stopping voltage corresponds to the electrons' maximum kinetic energy.
Stopping potential, `eV_0 = hν_"incident" - phi`
Where ν is the frequency of the incident radiation and Φ is the metal surface's work function. As a result, stopping potential rises as incident radiation frequency rises.
APPEARS IN
संबंधित प्रश्न
The photoelectric cut-off voltage in a certain experiment is 1.5 V. What is the maximum kinetic energy of photoelectrons emitted?
Ultraviolet light of wavelength 2271 Å from a 100 W mercury source irradiates a photo-cell made of molybdenum metal. If the stopping potential is −1.3 V, estimate the work function of the metal. How would the photo-cell respond to a high intensity (∼105 W m−2) red light of wavelength 6328 Å produced by a He-Ne laser?
Every metal has a definite work function. Why do all photoelectrons not come out with the same energy if incident radiation is monochromatic? Why is there an energy distribution of photoelectrons?
Light of wavelength λ falls on a metal with work-function hc/λ0. Photoelectric effect will take place only if
Photoelectric effect supports quantum nature of light because
(a) there is a minimum frequency below which no photoelectrons are emitted
(b) the maximum kinetic energy of photoelectrons depends only on the frequency of light and not on its intensity
(c) even when the metal surface is faintly illuminated the photoelectrons leave the surface immediately
(d) electric charge of the photoelectrons is quantised
If the wavelength of light in an experiment on photoelectric effect is doubled,
(a) photoelectric emission will not take place
(b) photoelectric emission may or may not take place
(c) the stopping potential will increase
(d) the stopping potential will decrease
Define the terms "stopping potential' and 'threshold frequency' in relation to the photoelectric effect. How does one determine these physical quantities using Einstein's equation?
The graph shows the variation of photocurrent for a photosensitive metal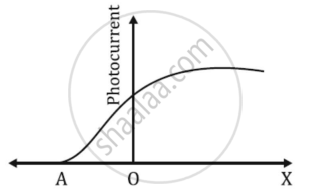
- What does X and A on the horizontal axis represent?
- Draw this graph for three different values of frequencies of incident radiation ʋ1, ʋ2 and ʋ3 (ʋ3 > ʋ2 > ʋ1) for the same intensity.
- Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I3 > I2 > I1) having the same frequency.
Why it is the frequency and not the intensity of the light source that determines whether the emission of photoelectrons will occur or not? Explain.
A metallic plate exposed to white light emits electrons. For which of the following colours of light, the stopping potential will be maximum?
