Advertisements
Advertisements
प्रश्न
What modification can you suggest in the Hardy-Schulze law?
उत्तर
Hardy-Schulze law states that ‘the greater the valence of the flocculating ion added, the greater is its power to cause precipitation.
This law takes into consideration only the charge carried by an ion, not its size. The smaller the size of an ion, the more will be its polarising power. Thus, Hardy-Schulze law can be modified in terms of the polarising power of the flocculating ion. Thus, the modified Hardy-Schulze law can be stated as ‘the greater the polarising power of the flocculating ion added, the greater is its power to cause precipitation.
APPEARS IN
संबंधित प्रश्न
What is the reason for the stability of colloidal sols?
Define the following term:
Zeta potential
Out of MgCl2 and AlCl3, which one is more effective in causing coagulation of negatively charged sol and why?
Out of BaCl2 and KCl, which one is more effective in causing coagulation of a negatively charged colloidal Sol? Give reason.
Explain what is observed When a beam of light is passed through a colloidal sol.
Explain the terms Coagulation
What happens when persistent dialysis of a colloidal solution is carried out?
What happens when an emulsion is centrifuged?
A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the solution represented?

A colloidal sol is prepared by the given method in the figure. What is the charge on hydrated ferric oxide colloidal particles formed in the test tube? How is the sol represented?
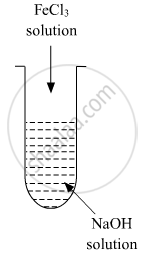
Which of the following statements are correct?
(i) Mixing two oppositely charged sols neutralises their charges and stabilises the colloid.
(ii) Presence of equal and similar charges on colloidal particles provides stability to the colloids.
(iii) Any amount of dispersed liquid can be added to emulsion without destabilising it.
(iv) Brownian movement stabilises sols.
How does it become possible to cause artificial rain by spraying silver iodide on the clouds?
Why do we add alum to purify water?
What causes brownian motion in colloidal dispersion?
Match the statement given in Column I with the phenomenon given in Column II.
| Column I | Column II |
| (i) Dispersion medium moves in an electric field | (a) Osmosis |
| (ii) Solvent molecules pass through semi permeable membrane towards solvent side |
(b) Electrophoresis |
| (iii) Movement of charged colloidal particles under the influence of applied electric potential towards oppositely charged electrodes |
(c) Electroosmosis |
| (iv) Solvent molecules pass through semi permeable membranes towards solution side |
(d) Reverse osmosis |
Assertion: Coagulation power of Al3+ is more than Na+.
Reason: Greater the valency of the flocculating ion added, greater is its power to cause precipitation (Hardy Schulze rule).
Gold number is associated with:-
Gold number of protective colloids A, Band Dare 0.50, 0.01 and 0.10 and 0.005. The correct order of their protecting power is
Lyophilic sols are more stable than lyophobic sols because ______.
