Advertisements
Advertisements
प्रश्न
What would be the mass of CO2 occupying a volume of 44 litres at 25°C and 750 mm pressure.
उत्तर
P1 = 750 mm P2 = 760mm
V1 = 44 lit. V2 = ?
T1 = 298K T2 = 273K
`("P"_1"V"_1)/"T"_1 = ("P"_2"V"_2)/"T"_2`
V2 = `("P"_1"V"_1"T"_2)/("T"_1"P"_2)`
= `(750 xx 44 xx 273)/(298 xx 760)`
∴ V2 = 39.78 lit.
22.4 lit. of CO2 at STP has mass = 44 g
∴ 39.78 lit. of CO2 at STP has mass = `(44 xx 39.78)/22.4`
= 78.14 g
APPEARS IN
संबंधित प्रश्न
What is meant by aqueous tension? How is the pressure exerted by a gas corrected to account for aqueous tension?
Convert −100° C to kelvin
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
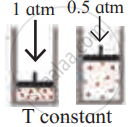 |
______________ |
With the help of the graph answer the following -

At constant temperature, Identify the law.
Use of hot air balloon in sports and meteorological observation is an application of
Assertion: Critical temperature of CO2 is 304 K, it can be liquefied above 304 K.
Reason: For a given mass of gas, volume is to directly proportional to pressure at constant temperature
Name two items that can serve as a model for Gay Lusaac’s law and explain.
Explain the following observation.
The size of a weather balloon becomes larger and larger as it ascends up to larger altitude
A certain mass of a gas occupies a volume of 2 dm3 at STP. At what temperature the volume of gas becomes double, keeping the pressure constant?
If 300 mL of a gas at 26.85°C is cooled to 6.85°C at constant pressure. What will be the final volume of gas?
