Advertisements
Advertisements
प्रश्न
Answer the following question.
When MnO2 is fused with KOH in the presence of KNO3 as an oxidizing agent, it gives a dark green compound (A). Compound (A) disproportionates in an acidic solution to give a purple compound (B). An alkaline solution of compound (B) oxidizes KI to compound (C) whereas an acidified solution of compound (B) oxidizes KI to (D). Identify (A), (B), (C), and (D).
उत्तर
Potassium permanganate is prepared by fusion of MnO2 with an alkali metal hydroxide and an oxidizing agent like KNO3. This produces the dark green K2MnO4 which disproportionates in a neutral or acidic solution to give permanganate.
\[\ce{2MnO2 + 4KOH + O2 -> \underset{\text{(A) Dark Green}}{2K2MnO4}+ 2H2O}\]
\[\ce{3MnO^2-_4 + 4H^+ ->\underset{\text{(B) purple}}{2MnO^-_4} + MnO2 + 2H2O}\]
In acid solutions:
Iodine is librated from potassium iodine:
\[\ce{10I^- +\underset{\text{(B)}}{2MnO^-_4}+ 16H^+ -> 2Mn^2+ + 8H2O +\underset{\text{(D)}}{5l2}}\]
In neutral or faintly alkaline solution:
A notable reaction is the oxidation of iodine to iodate:
\[\ce{\underset{\text{(B)}}{2MnO^-_4}+ H2O + I^- -> 2MnO2 + 2OH^- + \underset{\text{(C)}}{IO^-_3}}\].
संबंधित प्रश्न
Complete the following equations:
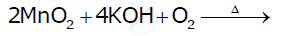
Using IUPAC norms write the formulae of Potassium trioxalatochromate (III)
On addition of small amount of \[\ce{KMnO4}\] to concentrated \[\ce{H2SO4}\], a green oily compound is obtained which is highly explosive in nature. Identify the compound from the following.
KMnO4 is coloured due to ______.
Why \[\ce{HCl}\] should not be used for potassium permanganate titrations?
What is the effect of pH on dichromate ion solution?
Potassium dichromate when heated with concentrated sulphuric acid and a soluble chloride, gives brown-red vapours of ______.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Describe the preparation of potassium permanganate. How does the acidified permanganate solution react with SO2? Write the ionic equation for the reaction.
Describe the preparation of potassium permanganate. How does the acidified permanganate solution react with oxalic acid? Write the ionic equation for the reaction.
