Advertisements
Advertisements
प्रश्न
When you exercise in the morning, by considering your body as a thermodynamic system, which of the following is true?
विकल्प
ΔU > 0, W > 0
ΔU < 0, W > 0
ΔU < 0, W < 0
ΔU = 0, W > 0
उत्तर
ΔU < 0, W > 0
APPEARS IN
संबंधित प्रश्न
Explain the cyclic process.
3 mole of a gas at temperature 400 K expands isothermally from an initial volume of 4 litres to a final volume of 8 litres. Find the work done by the gas. (R = 8.31 J mol-1 K-1)
Give an expression for work done in an isothermal process.
Give an equation state for an isochoric process.
Can the given heat energy be completely converted to work in a cyclic process? If not, when can the heat can completely converted to work?
Derive the work done in an isothermal process.
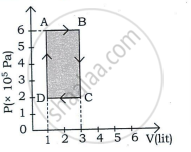
An ideal gas is taken in a cyclic process as shown in the figure. Calculate
- work done by the gas
- work done on the gas
- Net work done in the process

In an isothermal process, the volume of an ideal gas is halved. One can say that ____________.
In a certain thermodynamical process, the pressure of a gas depends on its volume as kV3. The work done when the temperature changes from 100°C to 300°C will be ______ nR, where n denotes number of moles of a gas.
An ideal gas is taken through a cyclic process ABCDA as shown in figure. The net work done by the gas during the cycle is ______.