Advertisements
Advertisements
प्रश्न
Why does cis-polyisoprene possess elastic property?
उत्तर
The cis-polyisoprene molecule consists of various chains held together by weak van der Waals interactions and has a coiled structure. Thus, it can be stretched like a spring and shows elastic properties.
APPEARS IN
संबंधित प्रश्न
Write the name of monomers used for getting the following polymers : Bakelite
Write the names of monomers used for getting the following polymers:
Teflon
Identify the monomer in the following polymeric structures.
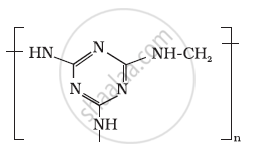
A natural linear polymer of 2-methyl-1, 3-butadiene becomes hard on treatment with sulphur between 373 to 415 K and – S – S – bonds are formed between chains. Write the structure of the product of this treatment?
To have practical applications why are cross links required in rubber?
What is the structural difference between HDP and LDP? How does the structure account for different behaviour and nature, hence the use of a polymer?
Assertion: Network polymers are thermosetting.
Reason: Network polymers have high molecular mass.
Which of the following is a basic dye?
Which one is disperse dye?
Linear chain polymers have ______.
