Advertisements
Advertisements
Question
Why does cis-polyisoprene possess elastic property?
Solution
The cis-polyisoprene molecule consists of various chains held together by weak van der Waals interactions and has a coiled structure. Thus, it can be stretched like a spring and shows elastic properties.
APPEARS IN
RELATED QUESTIONS
Write the names and structures of the monomers of the following polymers: Polyvinyl chloride
Write the names of monomers used for getting the following polymers:
Teflon
Identify the monomer in the following polymeric structures.
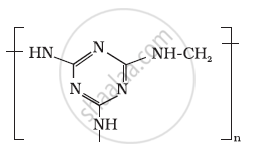
Write the names and structures of the monomers of the following polymers:
(i) Terylene
(ii) Bakelite
(iii) Buna-S
A natural linear polymer of 2-methyl-1, 3-butadiene becomes hard on treatment with sulphur between 373 to 415 K and – S – S – bonds are formed between chains. Write the structure of the product of this treatment?
Assertion: Network polymers are thermosetting.
Reason: Network polymers have high molecular mass.
Which of the following is a cross linked polymer?
Which of the following is a basic dye?
Which one is disperse dye?
Linear chain polymers have ______.
