Advertisements
Advertisements
प्रश्न
Why is the size of neon greater than fluorine?
उत्तर
The size of Neon is bigger compared to fluorine because the outer shell of neon is complete (octet). As a result, the effect of nuclear pull over the valence shell electrons cannot be seen. Hence the size of Neon is greater than fluorine.
संबंधित प्रश्न
Which is greater in size an atom or an anion?
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
Give scientific reasons for the following:
Atomic radius goes on decreasing while going from left to right in a period.
Arrange the following as per the instruction given in the bracket
Na, K, Li (Increasing atomic size)
The size of an atom is indicated by its _______.
Some elements and their atomic radii are given here. Arrange them in decreasing order of their atomic radii. Identify which of the above elements is the biggest atom and which is smallest?
| Element | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
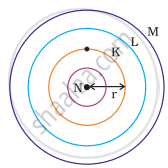
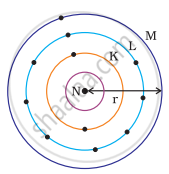
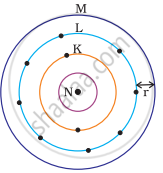
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
- In below ladder symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their atomic number in the Periodic Table.
- Arrange them in the order of their group also.

Which one of the following has the largest atomic radius?